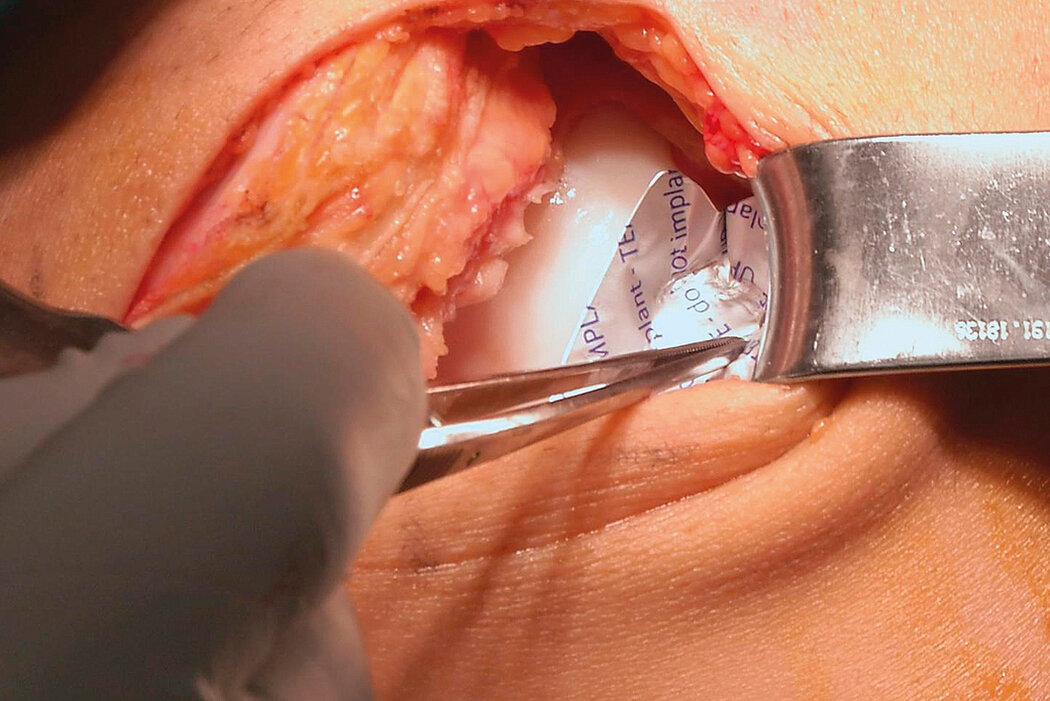
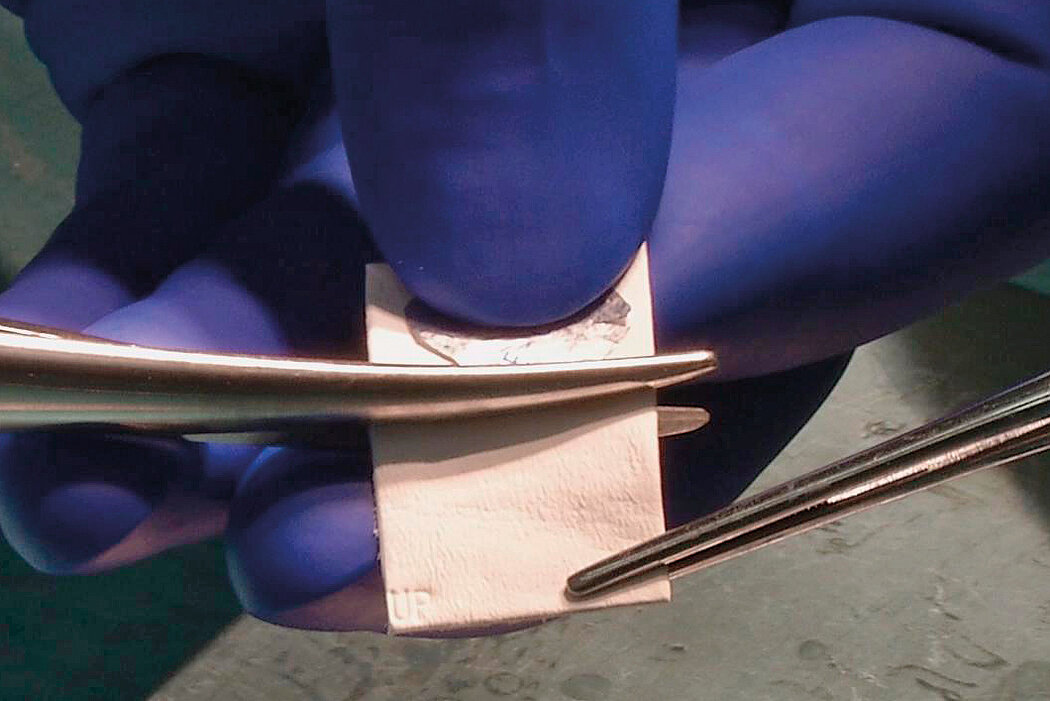
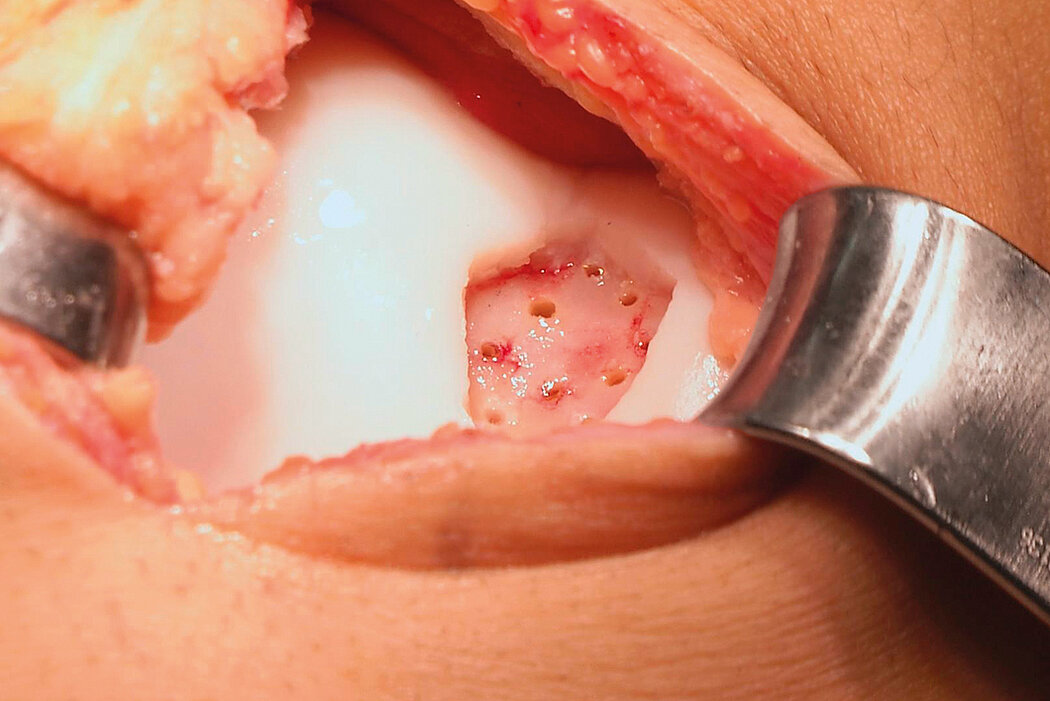
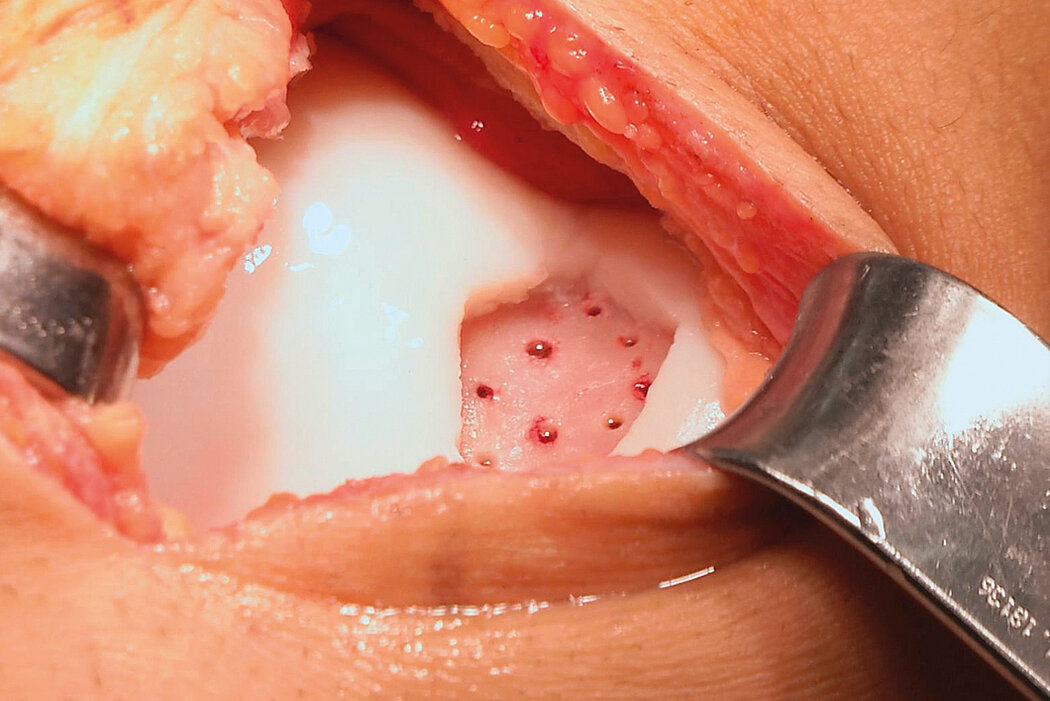
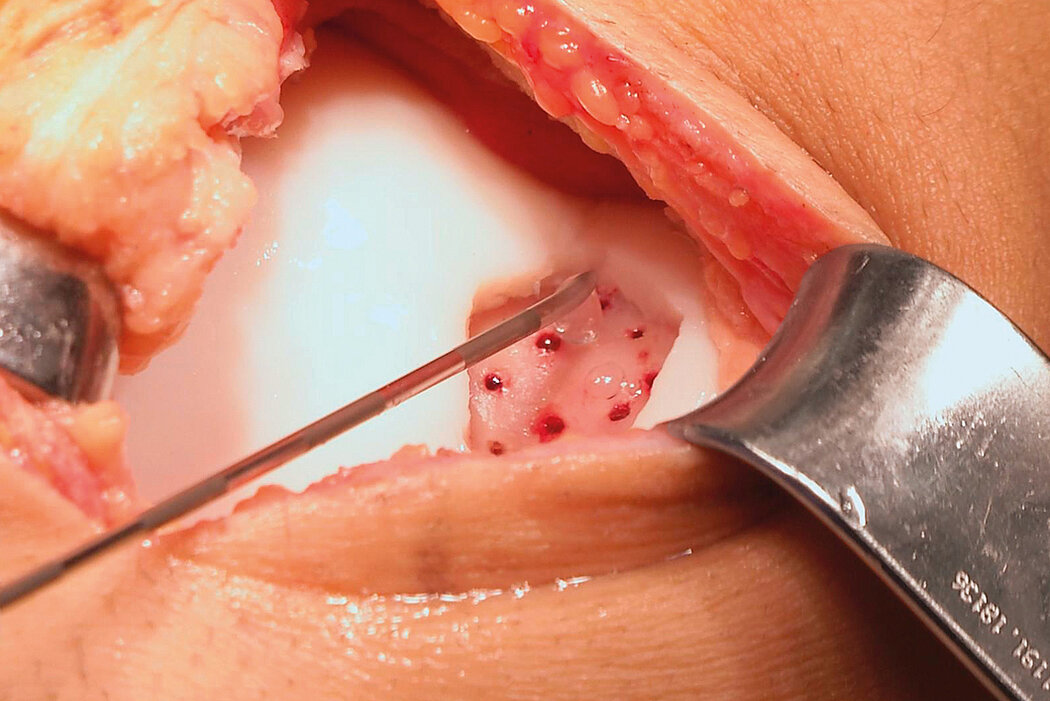
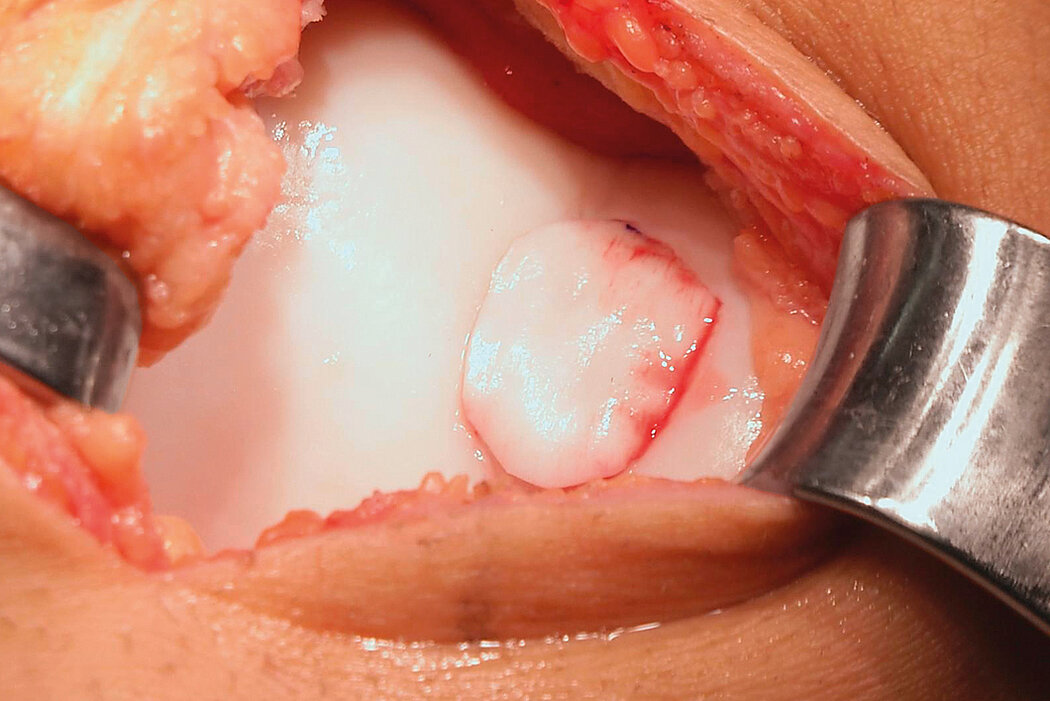
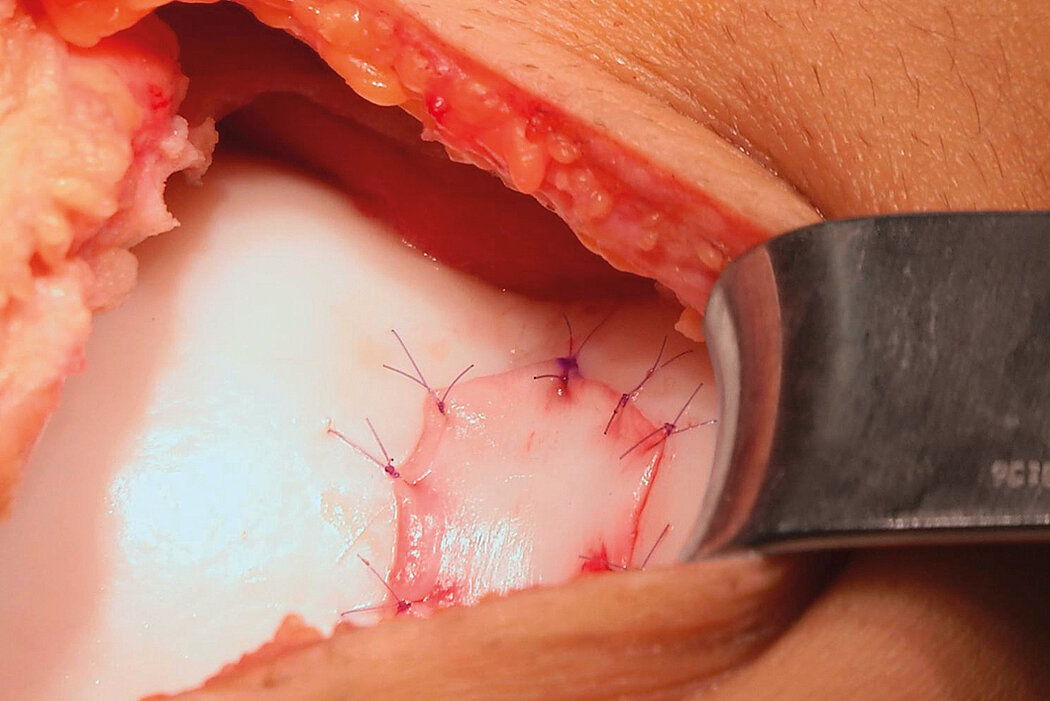
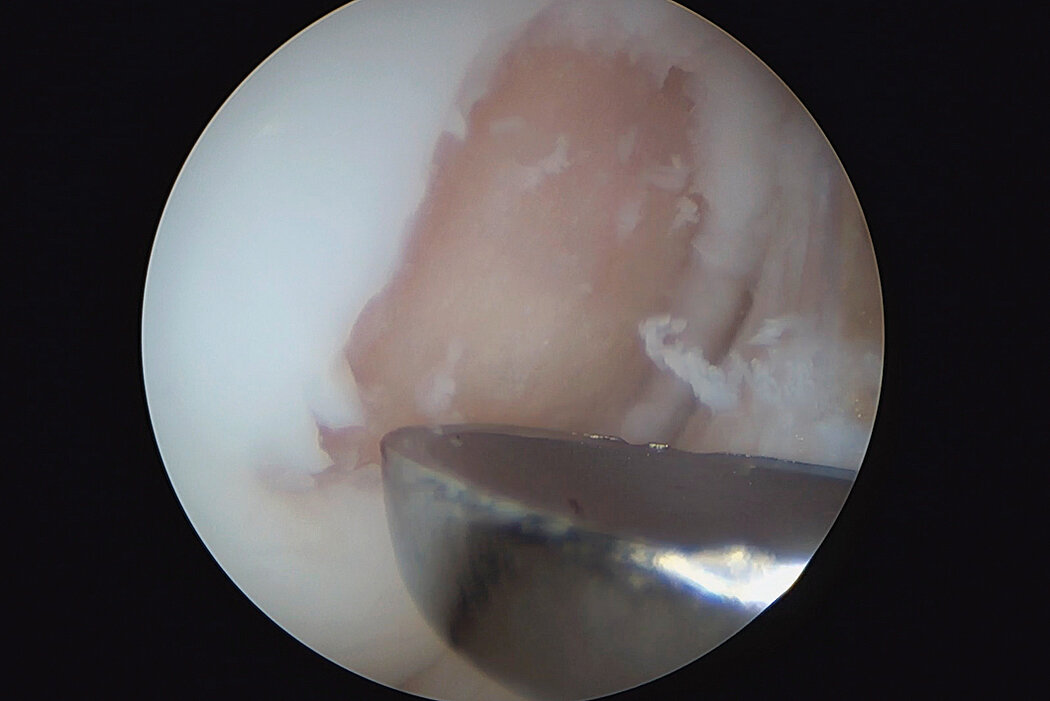
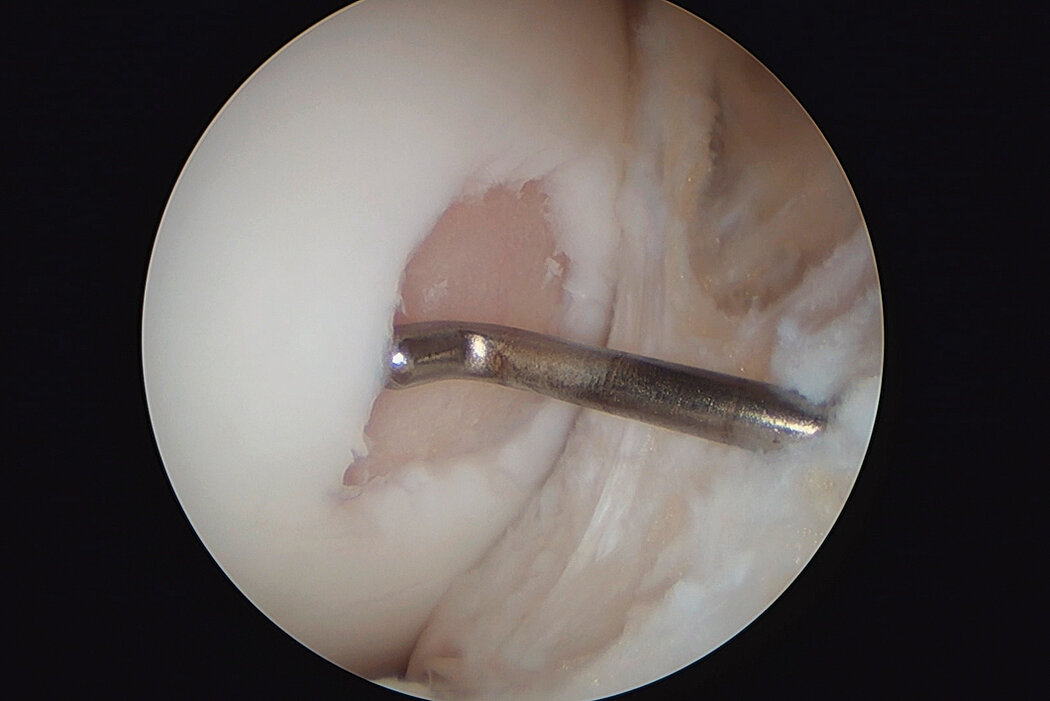
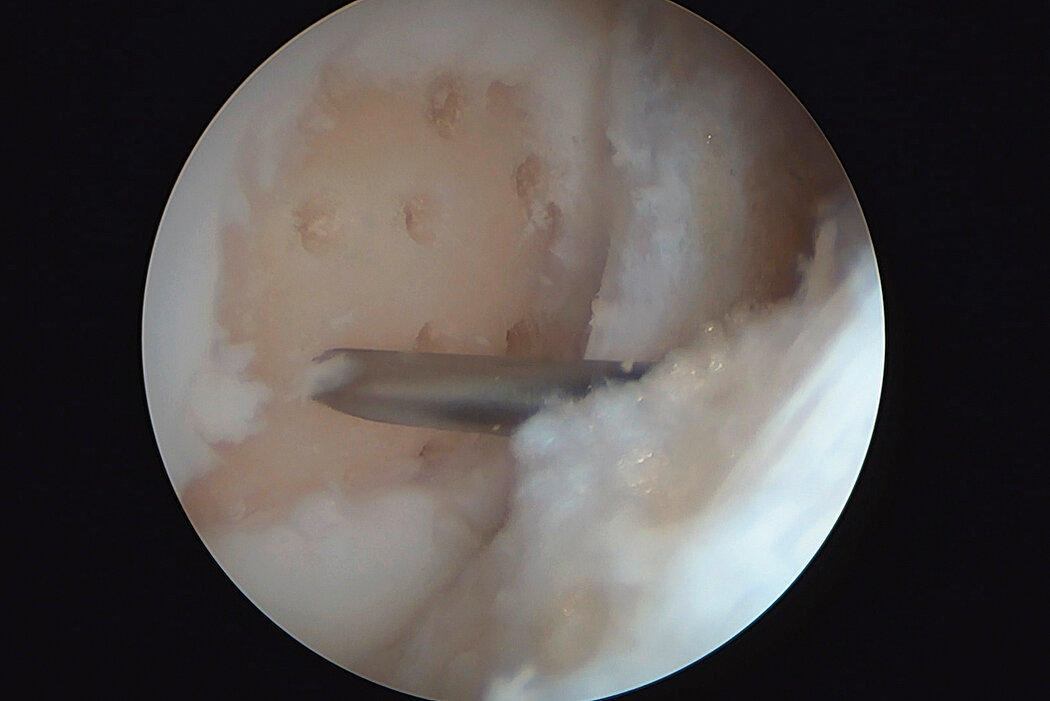
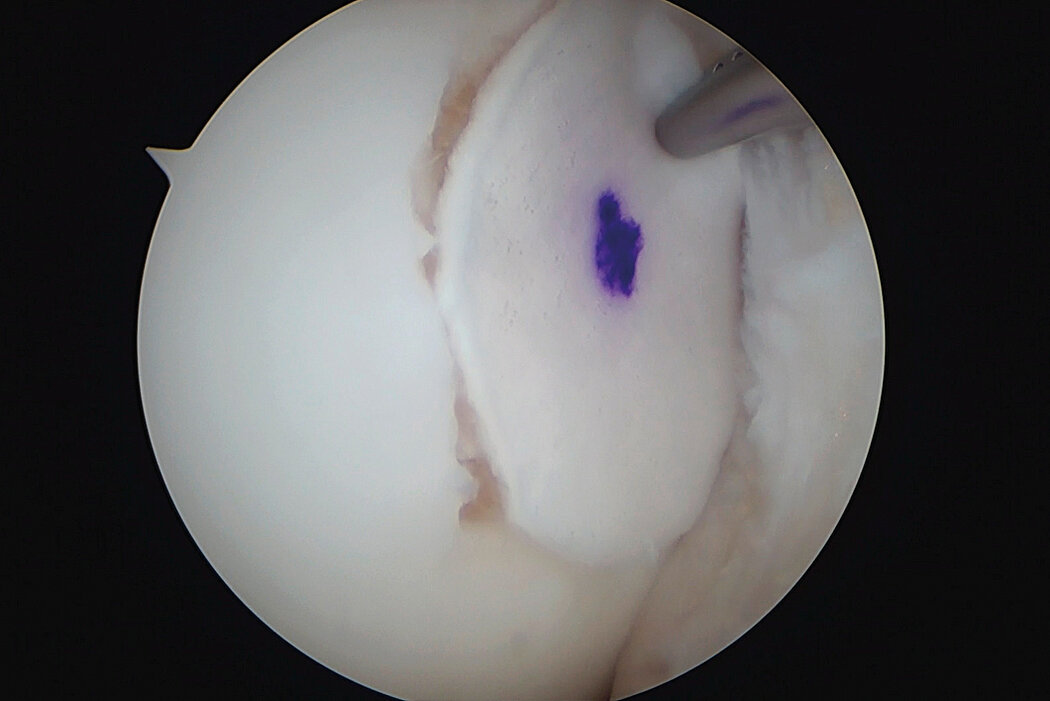
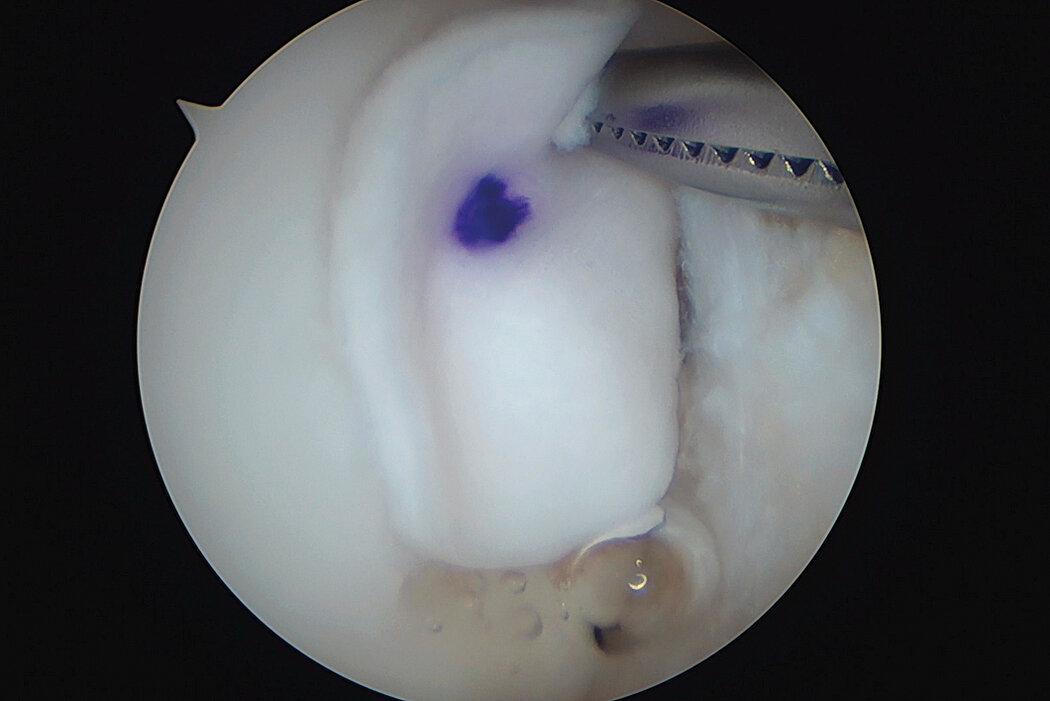
AMIC® Chondro-Gide® in the Knee

AMIC® Chondro-Gide® is a minimally invasive 1-step treatment that uses Autologous Matrix-Induced Chondrogenesis (AMIC®) combined with Chondro-Gide® to repair cartilage defects of all sizes. Backed by more than 10 years of clinical success, AMIC® Chondro-Gide® is an effective and cost-effective treatment1,2,3 for repairing damaged knee cartilage, alleviating or preventing pain, and slowing the progression of damage.
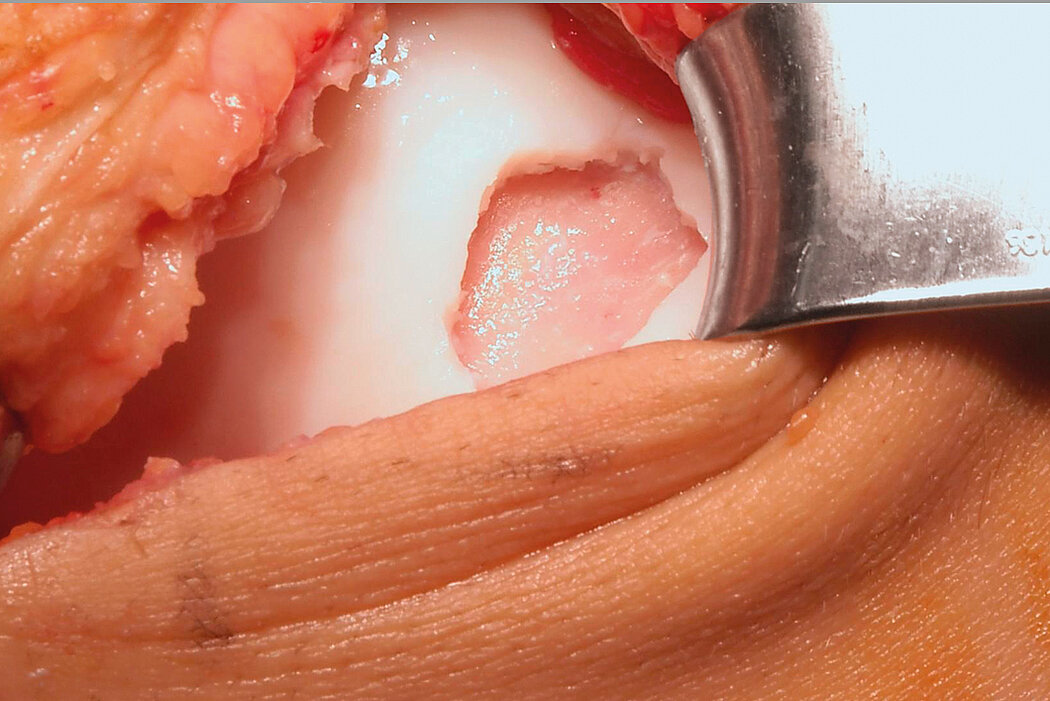
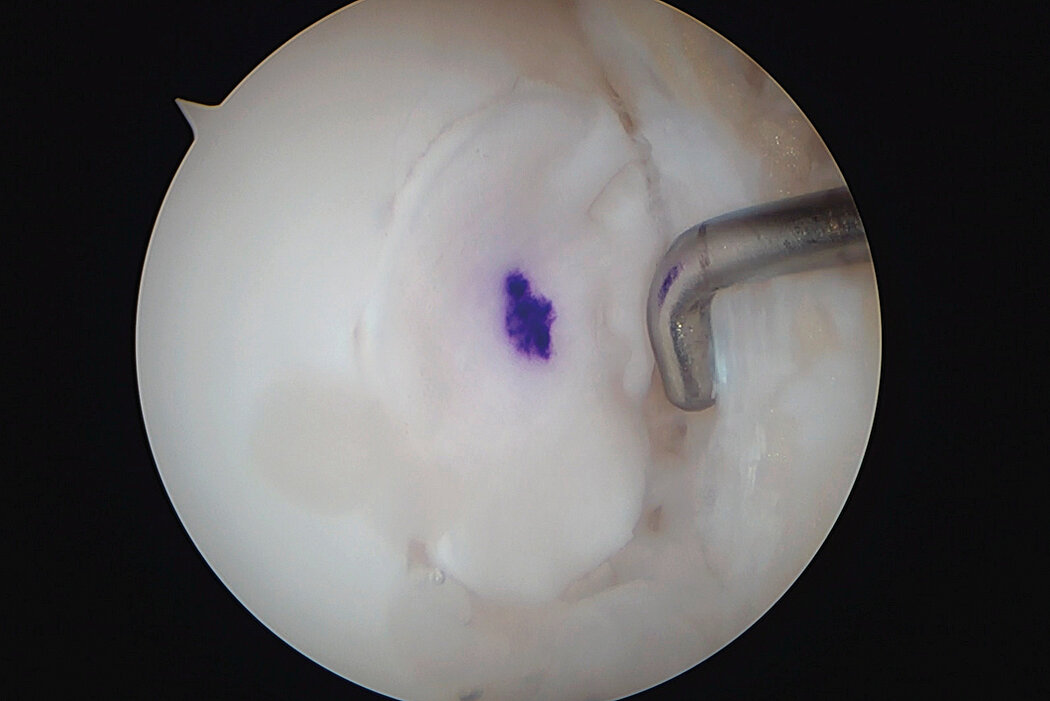
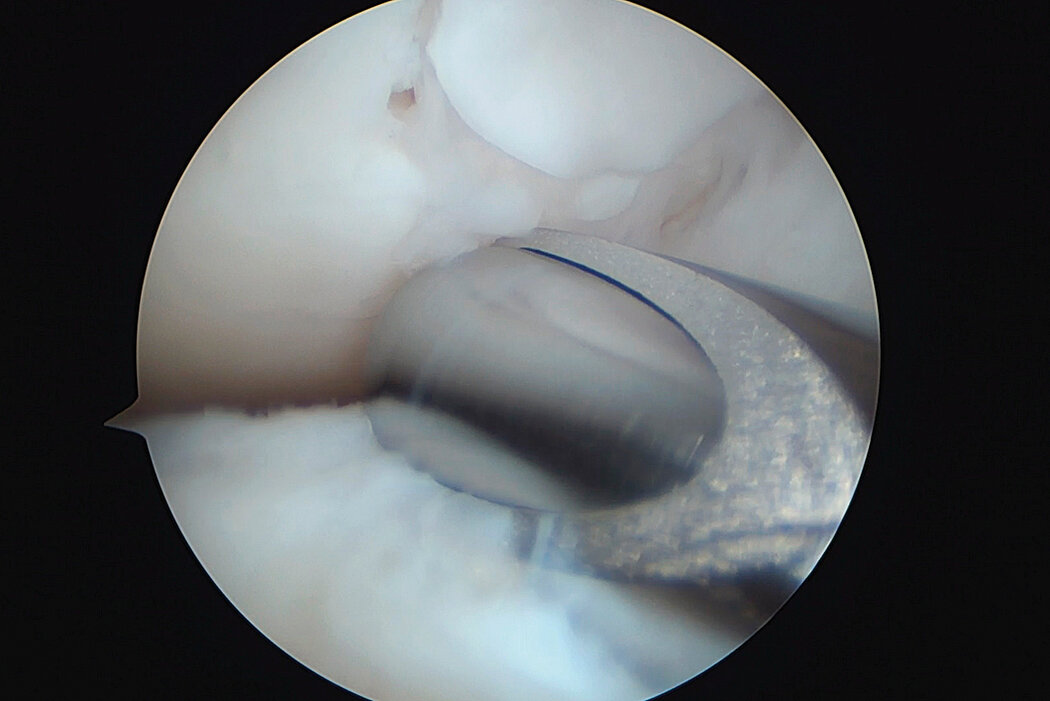
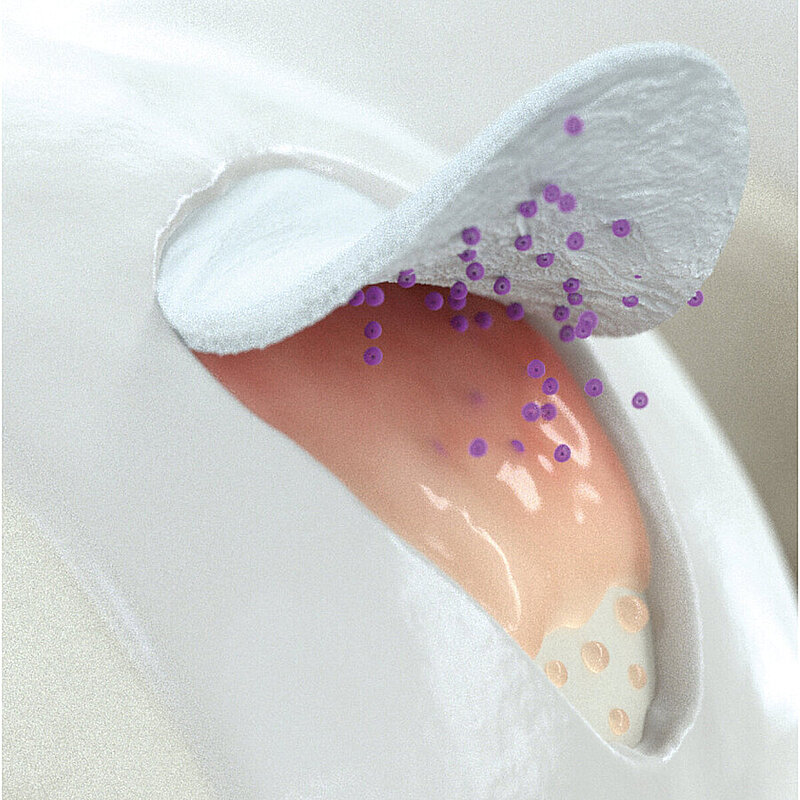
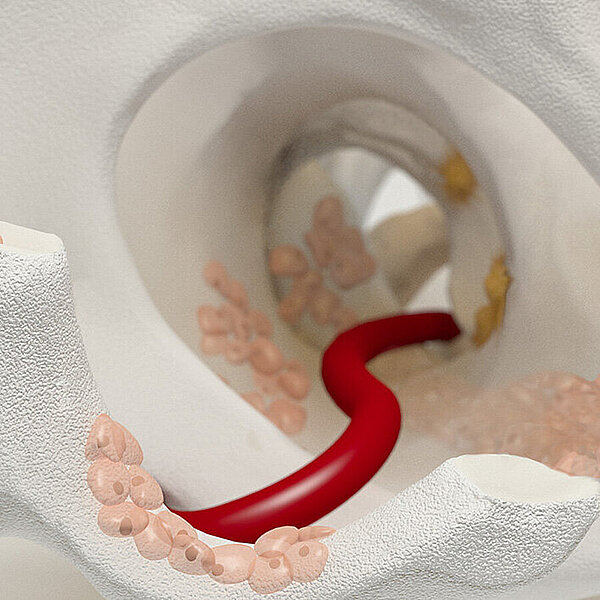
AMIC® Chondro-Gide® combines microfracturing (MFx) with the use of Chondro-Gide®, which covers and protects both the super clot resulting from MFx and the repair tissue.4 It can be performed either by mini-open or arthroscopic methods. Biocompatible and fully resorbable, Chondro-Gide® supports the body’s own healing potential.
Benefits
With its specially designed bilayer structure, Chondro-Gide® provides a protective environment that fosters the growth of new tissue.5,6
- Bio-derived, bilayer Collagen I/III membrane5
- Biocompatible and naturally resorbed5
- Easy to handle: supple and tear-resistant5
- Can be glued into place5
- Compatible with a range of tissue regeneration techniques7
- One-step procedure5
- Ready for use off the shelf5
References
- SCHIAVONE PANNI, A., et al. Good clinical results with autologous matrix-induced chondrogenesis (Amic) technique in large knee chondral defects. Knee Surg Sports Traumatol Arthrosc, 2018 Apr 26(4):1130-36 (Clinical study)
- WALTHER, M., et al. Scaffold based reconstruction of focal full thickness talar cartilage defects. Clinical Research on Foot & Ankle, 2013, 1-5. (Clinical study)
- KAISER, N., et al. Clinical results 10 years after AMIC in the knee. Swiss Med Wkly, 2015, 145 (Suppl 210), 43S. (Clinical study)
- VOLZ, M., et al. A randomized controlled trial demonstrating sustained benefit of Autologous Matrix-Induced Chondrogenesis over microfracture at five years. Int Orthop, Apr 2017, 41(4), 797-804. (Clinical study)
- Geistlich Pharma AG data on file (Bench test)
- GILLE, J., et al. Cell-Laden and Cell-Free Matrix-Induced-Chondrogenesis versus Microfracture for the Treatment of Articular Cartilage Defects: A Histological and Biomechanical Study in Sheep. Cartilage OnlineFirst, January 7, 2010, doi:10.1177/1947603509358721 (Pre-clinical study)
- KRAMER, J., et al. In vivo matrix-guided human mesenchymal stem cells. Cell Mol Life Sci, Mar 2006, 63(5), 616-626. (Clinical study)
- MITHOEFER, K., et al. The microfracture technique for the sustained benefit of Autologous Matrix-Induced Chondrogenesis over microfracture at five years. Int Orthop, Apr 2017, 41(4), 797-804. (Clinical study)
- GOYAL, D., et al. Evidence-based status of microfracture technique: a systematic review of level I and II studies. Arthroscopy, Sep 2013, 29(9), 1579-1588. (Review of clinical studies)
- FONTANA, A., et al. Sustained five-year benefit of autologous matrix-induced chondrogenesis for femoral acetabular impingement-induced chondral lesions compared with microfracture treatment. Bone Joint J, May 2015, 97-B(5), 628-635. (Clinical study)
- GAO, L., et al. Early loss of subchondral bone following microfracture is counteracted by bone marrow aspirate in a translational model of osteochondral repair. Nature Scientif-ic Reports, 2017, 7:45189, DOI: 10.1038/srep45189 (Pre-clinical study)
- FRANK, R.M., et al., Failure of Bone Marrow Stimulation Techniques, Sports Med Arthrosc Rev, 2017, 25 (1) (Review of clinical studies)
- STEADMAN, J.R., Microfracture Technique for Full-Thickness Chondral Defects: Technique and Clinical Results. Operative Techniques in Orthopaedics. 1997. 7(4), 300-304. (Clinical study)
- KAISER, N., et al. Clinical results 10 years after AMIC in the knee. Swiss Med Wkly, 2015, 145 (Suppl 210), 43S. (Clinical study)
- VOLZ, M., et al. A randomized controlled trial demonstrating sustained benefit of Autologous Matrix-Induced Chondrogenesis over microfracture at five years. Int Orthop, Apr 2017, 41(4), 797-804. (Clinical study)
- SCHAGEMANN, J., et al. Mid-term outcome of arthroscopic AMIC for the treatment of articular cartilage defects in the knee joint is equivalent to mini-open procedures. Arch Orthop Trauma Surg, Jan 22 2018. (Clinical study)
- STEINWACHS, M.R.,et al. Systematic Review and Meta-Analysis of the Clinical Evidence on the Use of Autologous Matrix-Induced Chondrogenesis in the Knee. Cartilage. 2019:1947603519870846. https://www.ncbi.nlm.nih.gov/ pubmed/31508990 (Review of clinical studies)
- FOSSUM, V., et al. Collagen-Covered Autologous Chondrocyte Implantation Versus Autologous Matrix-Induced Chondrogenesis: A Randomized Trial Comparing 2 Methods for Repair of Cartilage Defects of the Knee. Orthopaedic Journal of Sports Medicine. 019;7(9):2325967119868212. doi.org/10.1177/2325967119868212 (Clinical study)
References
- SCHIAVONE PANNI, A., et al. Good clinical results with autologous matrix-induced chondrogenesis (Amic) technique in large knee chondral defects. Knee Surg Sports Traumatol Arthrosc, 2018 Apr 26(4):1130-36 (Clinical study)
- WALTHER, M., et al. Scaffold based reconstruction of focal full thickness talar cartilage defects. Clinical Research on Foot & Ankle, 2013, 1-5. (Clinical study)
- KAISER, N., et al. Clinical results 10 years after AMIC in the knee. Swiss Med Wkly, 2015, 145 (Suppl 210), 43S. (Clinical study)
- VOLZ, M., et al. A randomized controlled trial demonstrating sustained benefit of Autologous Matrix-Induced Chondrogenesis over microfracture at five years. Int Orthop, Apr 2017, 41(4), 797-804. (Clinical study)
- Geistlich Pharma AG data on file (Bench test)
- GILLE, J., et al. Cell-Laden and Cell-Free Matrix-Induced-Chondrogenesis versus Microfracture for the Treatment of Articular Cartilage Defects: A Histological and Biomechanical Study in Sheep. Cartilage OnlineFirst, January 7, 2010, doi:10.1177/1947603509358721 (Pre-clinical study)
- KRAMER, J., et al. In vivo matrix-guided human mesenchymal stem cells. Cell Mol Life Sci, Mar 2006, 63(5), 616-626. (Clinical study)
- MITHOEFER, K., et al. The microfracture technique for the sustained benefit of Autologous Matrix-Induced Chondrogenesis over microfracture at five years. Int Orthop, Apr 2017, 41(4), 797-804. (Clinical study)
- GOYAL, D., et al. Evidence-based status of microfracture technique: a systematic review of level I and II studies. Arthroscopy, Sep 2013, 29(9), 1579-1588. (Review of clinical studies)
- FONTANA, A., et al. Sustained five-year benefit of autologous matrix-induced chondrogenesis for femoral acetabular impingement-induced chondral lesions compared with microfracture treatment. Bone Joint J, May 2015, 97-B(5), 628-635. (Clinical study)
- GAO, L., et al. Early loss of subchondral bone following microfracture is counteracted by bone marrow aspirate in a translational model of osteochondral repair. Nature Scientif-ic Reports, 2017, 7:45189, DOI: 10.1038/srep45189 (Pre-clinical study)
- FRANK, R.M., et al., Failure of Bone Marrow Stimulation Techniques, Sports Med Arthrosc Rev, 2017, 25 (1) (Review of clinical studies)
- STEADMAN, J.R., Microfracture Technique for Full-Thickness Chondral Defects: Technique and Clinical Results. Operative Techniques in Orthopaedics. 1997. 7(4), 300-304. (Clinical study)
- KAISER, N., et al. Clinical results 10 years after AMIC in the knee. Swiss Med Wkly, 2015, 145 (Suppl 210), 43S. (Clinical study)
- VOLZ, M., et al. A randomized controlled trial demonstrating sustained benefit of Autologous Matrix-Induced Chondrogenesis over microfracture at five years. Int Orthop, Apr 2017, 41(4), 797-804. (Clinical study)
- SCHAGEMANN, J., et al. Mid-term outcome of arthroscopic AMIC for the treatment of articular cartilage defects in the knee joint is equivalent to mini-open procedures. Arch Orthop Trauma Surg, Jan 22 2018. (Clinical study)
- STEINWACHS, M.R.,et al. Systematic Review and Meta-Analysis of the Clinical Evidence on the Use of Autologous Matrix-Induced Chondrogenesis in the Knee. Cartilage. 2019:1947603519870846. https://www.ncbi.nlm.nih.gov/ pubmed/31508990 (Review of clinical studies)
- FOSSUM, V., et al. Collagen-Covered Autologous Chondrocyte Implantation Versus Autologous Matrix-Induced Chondrogenesis: A Randomized Trial Comparing 2 Methods for Repair of Cartilage Defects of the Knee. Orthopaedic Journal of Sports Medicine. 019;7(9):2325967119868212. doi.org/10.1177/2325967119868212 (Clinical study)
Mini-Open Surgical Technique, Prof Matthias Steinwachs
References
- SCHIAVONE PANNI, A., et al. Good clinical results with autologous matrix-induced chondrogenesis (Amic) technique in large knee chondral defects. Knee Surg Sports Traumatol Arthrosc, 2018 Apr 26(4):1130-36 (Clinical study)
- WALTHER, M., et al. Scaffold based reconstruction of focal full thickness talar cartilage defects. Clinical Research on Foot & Ankle, 2013, 1-5. (Clinical study)
- KAISER, N., et al. Clinical results 10 years after AMIC in the knee. Swiss Med Wkly, 2015, 145 (Suppl 210), 43S. (Clinical study)
- VOLZ, M., et al. A randomized controlled trial demonstrating sustained benefit of Autologous Matrix-Induced Chondrogenesis over microfracture at five years. Int Orthop, Apr 2017, 41(4), 797-804. (Clinical study)
- Geistlich Pharma AG data on file (Bench test)
- GILLE, J., et al. Cell-Laden and Cell-Free Matrix-Induced-Chondrogenesis versus Microfracture for the Treatment of Articular Cartilage Defects: A Histological and Biomechanical Study in Sheep. Cartilage OnlineFirst, January 7, 2010, doi:10.1177/1947603509358721 (Pre-clinical study)
- KRAMER, J., et al. In vivo matrix-guided human mesenchymal stem cells. Cell Mol Life Sci, Mar 2006, 63(5), 616-626. (Clinical study)
- MITHOEFER, K., et al. The microfracture technique for the sustained benefit of Autologous Matrix-Induced Chondrogenesis over microfracture at five years. Int Orthop, Apr 2017, 41(4), 797-804. (Clinical study)
- GOYAL, D., et al. Evidence-based status of microfracture technique: a systematic review of level I and II studies. Arthroscopy, Sep 2013, 29(9), 1579-1588. (Review of clinical studies)
- FONTANA, A., et al. Sustained five-year benefit of autologous matrix-induced chondrogenesis for femoral acetabular impingement-induced chondral lesions compared with microfracture treatment. Bone Joint J, May 2015, 97-B(5), 628-635. (Clinical study)
- GAO, L., et al. Early loss of subchondral bone following microfracture is counteracted by bone marrow aspirate in a translational model of osteochondral repair. Nature Scientif-ic Reports, 2017, 7:45189, DOI: 10.1038/srep45189 (Pre-clinical study)
- FRANK, R.M., et al., Failure of Bone Marrow Stimulation Techniques, Sports Med Arthrosc Rev, 2017, 25 (1) (Review of clinical studies)
- STEADMAN, J.R., Microfracture Technique for Full-Thickness Chondral Defects: Technique and Clinical Results. Operative Techniques in Orthopaedics. 1997. 7(4), 300-304. (Clinical study)
- KAISER, N., et al. Clinical results 10 years after AMIC in the knee. Swiss Med Wkly, 2015, 145 (Suppl 210), 43S. (Clinical study)
- VOLZ, M., et al. A randomized controlled trial demonstrating sustained benefit of Autologous Matrix-Induced Chondrogenesis over microfracture at five years. Int Orthop, Apr 2017, 41(4), 797-804. (Clinical study)
- SCHAGEMANN, J., et al. Mid-term outcome of arthroscopic AMIC for the treatment of articular cartilage defects in the knee joint is equivalent to mini-open procedures. Arch Orthop Trauma Surg, Jan 22 2018. (Clinical study)
- STEINWACHS, M.R.,et al. Systematic Review and Meta-Analysis of the Clinical Evidence on the Use of Autologous Matrix-Induced Chondrogenesis in the Knee. Cartilage. 2019:1947603519870846. https://www.ncbi.nlm.nih.gov/ pubmed/31508990 (Review of clinical studies)
- FOSSUM, V., et al. Collagen-Covered Autologous Chondrocyte Implantation Versus Autologous Matrix-Induced Chondrogenesis: A Randomized Trial Comparing 2 Methods for Repair of Cartilage Defects of the Knee. Orthopaedic Journal of Sports Medicine. 019;7(9):2325967119868212. doi.org/10.1177/2325967119868212 (Clinical study)
Arthroscopic Surgical Technique, Prof Justus Gille
References
- SCHIAVONE PANNI, A., et al. Good clinical results with autologous matrix-induced chondrogenesis (Amic) technique in large knee chondral defects. Knee Surg Sports Traumatol Arthrosc, 2018 Apr 26(4):1130-36 (Clinical study)
- WALTHER, M., et al. Scaffold based reconstruction of focal full thickness talar cartilage defects. Clinical Research on Foot & Ankle, 2013, 1-5. (Clinical study)
- KAISER, N., et al. Clinical results 10 years after AMIC in the knee. Swiss Med Wkly, 2015, 145 (Suppl 210), 43S. (Clinical study)
- VOLZ, M., et al. A randomized controlled trial demonstrating sustained benefit of Autologous Matrix-Induced Chondrogenesis over microfracture at five years. Int Orthop, Apr 2017, 41(4), 797-804. (Clinical study)
- Geistlich Pharma AG data on file (Bench test)
- GILLE, J., et al. Cell-Laden and Cell-Free Matrix-Induced-Chondrogenesis versus Microfracture for the Treatment of Articular Cartilage Defects: A Histological and Biomechanical Study in Sheep. Cartilage OnlineFirst, January 7, 2010, doi:10.1177/1947603509358721 (Pre-clinical study)
- KRAMER, J., et al. In vivo matrix-guided human mesenchymal stem cells. Cell Mol Life Sci, Mar 2006, 63(5), 616-626. (Clinical study)
- MITHOEFER, K., et al. The microfracture technique for the sustained benefit of Autologous Matrix-Induced Chondrogenesis over microfracture at five years. Int Orthop, Apr 2017, 41(4), 797-804. (Clinical study)
- GOYAL, D., et al. Evidence-based status of microfracture technique: a systematic review of level I and II studies. Arthroscopy, Sep 2013, 29(9), 1579-1588. (Review of clinical studies)
- FONTANA, A., et al. Sustained five-year benefit of autologous matrix-induced chondrogenesis for femoral acetabular impingement-induced chondral lesions compared with microfracture treatment. Bone Joint J, May 2015, 97-B(5), 628-635. (Clinical study)
- GAO, L., et al. Early loss of subchondral bone following microfracture is counteracted by bone marrow aspirate in a translational model of osteochondral repair. Nature Scientif-ic Reports, 2017, 7:45189, DOI: 10.1038/srep45189 (Pre-clinical study)
- FRANK, R.M., et al., Failure of Bone Marrow Stimulation Techniques, Sports Med Arthrosc Rev, 2017, 25 (1) (Review of clinical studies)
- STEADMAN, J.R., Microfracture Technique for Full-Thickness Chondral Defects: Technique and Clinical Results. Operative Techniques in Orthopaedics. 1997. 7(4), 300-304. (Clinical study)
- KAISER, N., et al. Clinical results 10 years after AMIC in the knee. Swiss Med Wkly, 2015, 145 (Suppl 210), 43S. (Clinical study)
- VOLZ, M., et al. A randomized controlled trial demonstrating sustained benefit of Autologous Matrix-Induced Chondrogenesis over microfracture at five years. Int Orthop, Apr 2017, 41(4), 797-804. (Clinical study)
- SCHAGEMANN, J., et al. Mid-term outcome of arthroscopic AMIC for the treatment of articular cartilage defects in the knee joint is equivalent to mini-open procedures. Arch Orthop Trauma Surg, Jan 22 2018. (Clinical study)
- STEINWACHS, M.R.,et al. Systematic Review and Meta-Analysis of the Clinical Evidence on the Use of Autologous Matrix-Induced Chondrogenesis in the Knee. Cartilage. 2019:1947603519870846. https://www.ncbi.nlm.nih.gov/ pubmed/31508990 (Review of clinical studies)
- FOSSUM, V., et al. Collagen-Covered Autologous Chondrocyte Implantation Versus Autologous Matrix-Induced Chondrogenesis: A Randomized Trial Comparing 2 Methods for Repair of Cartilage Defects of the Knee. Orthopaedic Journal of Sports Medicine. 019;7(9):2325967119868212. doi.org/10.1177/2325967119868212 (Clinical study)
Clinical Evidence AMIC® Knee
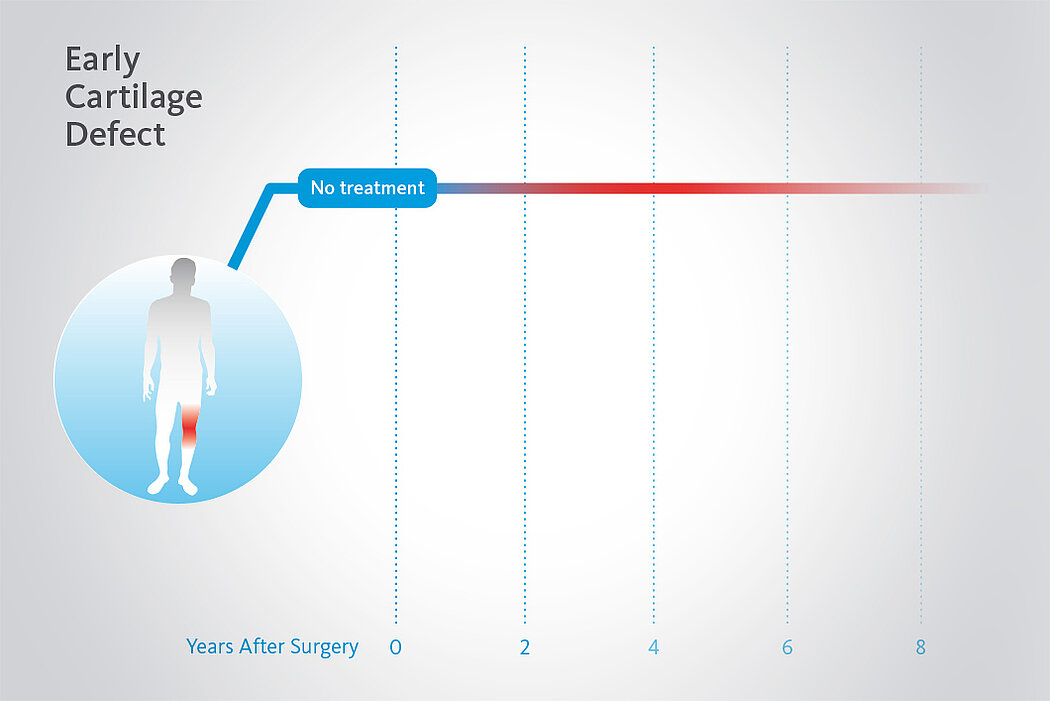
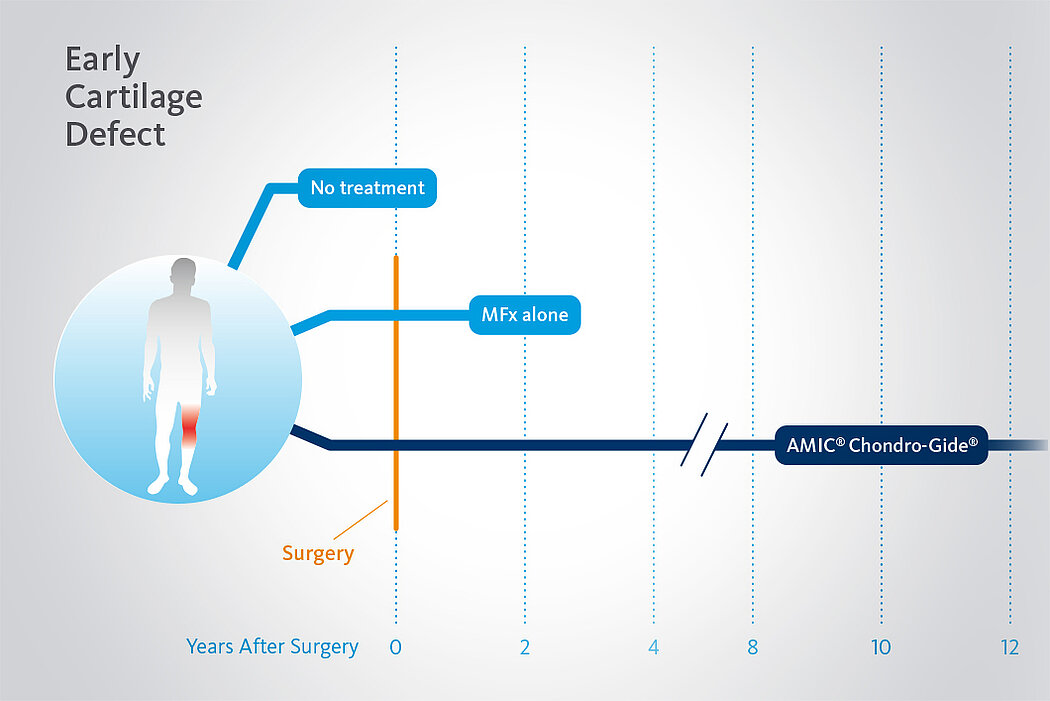
AMIC® Chondro-Gide® has been shown to be an effective treatment to repair cartilage lesions of all sizes in the knee. Clinical studies show successful treatment using AMIC® Chondro-Gide® with both mini-open and arthroscopic techniques for more than 10 years.14
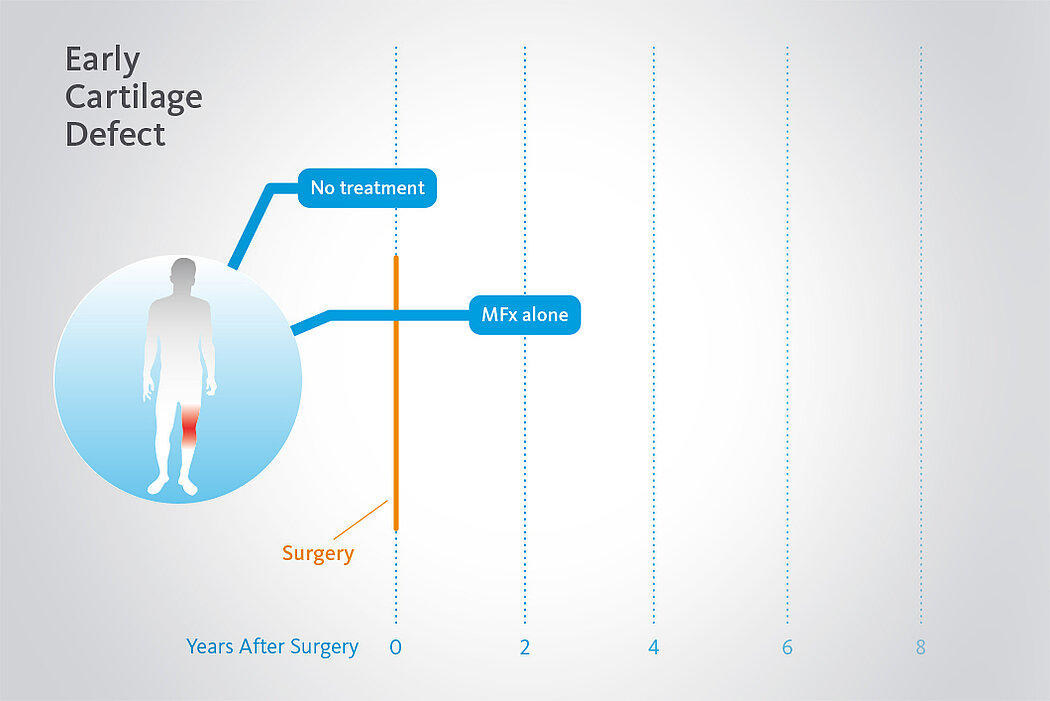
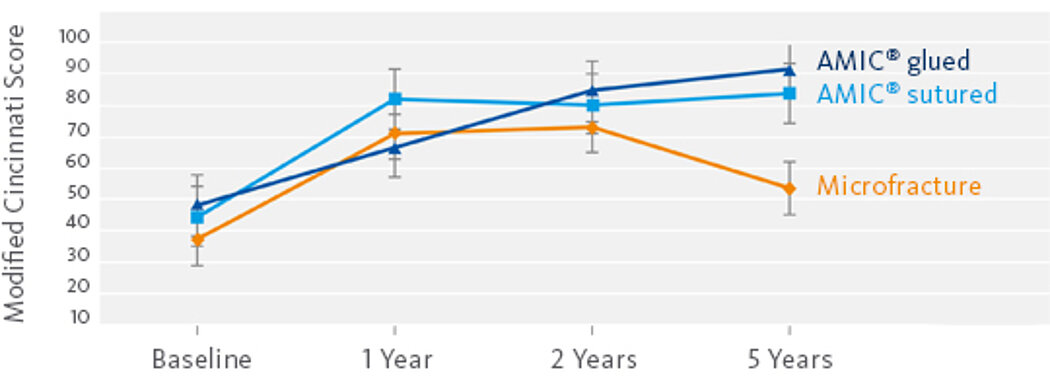
Better Performance than MFx Alone, After 2 Years
In a randomized, controlled 3-arm study of 47 patients in 2 centers15, a significant deterioration in results was seen after 2 years following MFx alone (without Chondro-Gide®). Pain and function scores (ICRS and modified Cincinnati scores) remained stable or even improved with AMIC®, while pain and function scores for the MFx group decreased.
References
- SCHIAVONE PANNI, A., et al. Good clinical results with autologous matrix-induced chondrogenesis (Amic) technique in large knee chondral defects. Knee Surg Sports Traumatol Arthrosc, 2018 Apr 26(4):1130-36 (Clinical study)
- WALTHER, M., et al. Scaffold based reconstruction of focal full thickness talar cartilage defects. Clinical Research on Foot & Ankle, 2013, 1-5. (Clinical study)
- KAISER, N., et al. Clinical results 10 years after AMIC in the knee. Swiss Med Wkly, 2015, 145 (Suppl 210), 43S. (Clinical study)
- VOLZ, M., et al. A randomized controlled trial demonstrating sustained benefit of Autologous Matrix-Induced Chondrogenesis over microfracture at five years. Int Orthop, Apr 2017, 41(4), 797-804. (Clinical study)
- Geistlich Pharma AG data on file (Bench test)
- GILLE, J., et al. Cell-Laden and Cell-Free Matrix-Induced-Chondrogenesis versus Microfracture for the Treatment of Articular Cartilage Defects: A Histological and Biomechanical Study in Sheep. Cartilage OnlineFirst, January 7, 2010, doi:10.1177/1947603509358721 (Pre-clinical study)
- KRAMER, J., et al. In vivo matrix-guided human mesenchymal stem cells. Cell Mol Life Sci, Mar 2006, 63(5), 616-626. (Clinical study)
- MITHOEFER, K., et al. The microfracture technique for the sustained benefit of Autologous Matrix-Induced Chondrogenesis over microfracture at five years. Int Orthop, Apr 2017, 41(4), 797-804. (Clinical study)
- GOYAL, D., et al. Evidence-based status of microfracture technique: a systematic review of level I and II studies. Arthroscopy, Sep 2013, 29(9), 1579-1588. (Review of clinical studies)
- FONTANA, A., et al. Sustained five-year benefit of autologous matrix-induced chondrogenesis for femoral acetabular impingement-induced chondral lesions compared with microfracture treatment. Bone Joint J, May 2015, 97-B(5), 628-635. (Clinical study)
- GAO, L., et al. Early loss of subchondral bone following microfracture is counteracted by bone marrow aspirate in a translational model of osteochondral repair. Nature Scientif-ic Reports, 2017, 7:45189, DOI: 10.1038/srep45189 (Pre-clinical study)
- FRANK, R.M., et al., Failure of Bone Marrow Stimulation Techniques, Sports Med Arthrosc Rev, 2017, 25 (1) (Review of clinical studies)
- STEADMAN, J.R., Microfracture Technique for Full-Thickness Chondral Defects: Technique and Clinical Results. Operative Techniques in Orthopaedics. 1997. 7(4), 300-304. (Clinical study)
- KAISER, N., et al. Clinical results 10 years after AMIC in the knee. Swiss Med Wkly, 2015, 145 (Suppl 210), 43S. (Clinical study)
- VOLZ, M., et al. A randomized controlled trial demonstrating sustained benefit of Autologous Matrix-Induced Chondrogenesis over microfracture at five years. Int Orthop, Apr 2017, 41(4), 797-804. (Clinical study)
- SCHAGEMANN, J., et al. Mid-term outcome of arthroscopic AMIC for the treatment of articular cartilage defects in the knee joint is equivalent to mini-open procedures. Arch Orthop Trauma Surg, Jan 22 2018. (Clinical study)
- STEINWACHS, M.R.,et al. Systematic Review and Meta-Analysis of the Clinical Evidence on the Use of Autologous Matrix-Induced Chondrogenesis in the Knee. Cartilage. 2019:1947603519870846. https://www.ncbi.nlm.nih.gov/ pubmed/31508990 (Review of clinical studies)
- FOSSUM, V., et al. Collagen-Covered Autologous Chondrocyte Implantation Versus Autologous Matrix-Induced Chondrogenesis: A Randomized Trial Comparing 2 Methods for Repair of Cartilage Defects of the Knee. Orthopaedic Journal of Sports Medicine. 019;7(9):2325967119868212. doi.org/10.1177/2325967119868212 (Clinical study)
Arthroscopic or Mini-Arthrotomy, both Give Equally Good Results
In a retrospective study, Schagemann et al.16 compared the clinical outcomes of AMIC® Chondro-Gide® procedures that were performed as arthroscopic or mini-open surgeries. The study followed patients up to 2 years. According to the patients’ Visual Analog Scale (VAS), Lysholm scores, and Knee injury and Osteoarthrithis Outcome Scores (KOOS), both surgical approaches yielded equally positive results.
First Meta-Analysis of 12 AMIC® Chondro-Gide® Studies Including 375 Patients
Most recently, (2019) in a systematic review and meta-analysis of AMIC® outcomes, the authors evaluated grade III/IV chondral and osteochondral lesions in the knee with a mean defect size of 4.24 cm2 with up to 7 years follow-up.
The use of AMIC® Chondro-Gide® in defect sizes, which are above the recommended threshold for MFx, significantly reduced pain and improved function from baseline to follow-up. The meta-analysis documents that the AMIC® Chondro-Gide® procedure significantly improves pain and functional outcomes in knee joints with chondral or osteochondral lesions. The results further demonstrate that the improvement was maintained over more than 5 years, which confirms the long-term success of AMIC® Chondro-Gide® in larger (>4 cm2) grade III and IV lesions.17
ACI-C and AMIC® Chondro-Gide® provide equally good results after 2 years
Fossum, et al., (2019) conducted a prospective, randomized, controlled study to assess the outcomes of ACI-C and AMIC® in chondral and osteochondral defects of the distal femur and patella. No significant differences were seen between the outcomes of the ACI-C and AMIC® techniques at 1 and 2 years.
The authors concluded that AMIC® could be considered a clinically equal, but less expensive alternative to ACI-C, as AMIC® is a 1-step procedure therefore far less resource-intensive.18
References
- SCHIAVONE PANNI, A., et al. Good clinical results with autologous matrix-induced chondrogenesis (Amic) technique in large knee chondral defects. Knee Surg Sports Traumatol Arthrosc, 2018 Apr 26(4):1130-36 (Clinical study)
- WALTHER, M., et al. Scaffold based reconstruction of focal full thickness talar cartilage defects. Clinical Research on Foot & Ankle, 2013, 1-5. (Clinical study)
- KAISER, N., et al. Clinical results 10 years after AMIC in the knee. Swiss Med Wkly, 2015, 145 (Suppl 210), 43S. (Clinical study)
- VOLZ, M., et al. A randomized controlled trial demonstrating sustained benefit of Autologous Matrix-Induced Chondrogenesis over microfracture at five years. Int Orthop, Apr 2017, 41(4), 797-804. (Clinical study)
- Geistlich Pharma AG data on file (Bench test)
- GILLE, J., et al. Cell-Laden and Cell-Free Matrix-Induced-Chondrogenesis versus Microfracture for the Treatment of Articular Cartilage Defects: A Histological and Biomechanical Study in Sheep. Cartilage OnlineFirst, January 7, 2010, doi:10.1177/1947603509358721 (Pre-clinical study)
- KRAMER, J., et al. In vivo matrix-guided human mesenchymal stem cells. Cell Mol Life Sci, Mar 2006, 63(5), 616-626. (Clinical study)
- MITHOEFER, K., et al. The microfracture technique for the sustained benefit of Autologous Matrix-Induced Chondrogenesis over microfracture at five years. Int Orthop, Apr 2017, 41(4), 797-804. (Clinical study)
- GOYAL, D., et al. Evidence-based status of microfracture technique: a systematic review of level I and II studies. Arthroscopy, Sep 2013, 29(9), 1579-1588. (Review of clinical studies)
- FONTANA, A., et al. Sustained five-year benefit of autologous matrix-induced chondrogenesis for femoral acetabular impingement-induced chondral lesions compared with microfracture treatment. Bone Joint J, May 2015, 97-B(5), 628-635. (Clinical study)
- GAO, L., et al. Early loss of subchondral bone following microfracture is counteracted by bone marrow aspirate in a translational model of osteochondral repair. Nature Scientif-ic Reports, 2017, 7:45189, DOI: 10.1038/srep45189 (Pre-clinical study)
- FRANK, R.M., et al., Failure of Bone Marrow Stimulation Techniques, Sports Med Arthrosc Rev, 2017, 25 (1) (Review of clinical studies)
- STEADMAN, J.R., Microfracture Technique for Full-Thickness Chondral Defects: Technique and Clinical Results. Operative Techniques in Orthopaedics. 1997. 7(4), 300-304. (Clinical study)
- KAISER, N., et al. Clinical results 10 years after AMIC in the knee. Swiss Med Wkly, 2015, 145 (Suppl 210), 43S. (Clinical study)
- VOLZ, M., et al. A randomized controlled trial demonstrating sustained benefit of Autologous Matrix-Induced Chondrogenesis over microfracture at five years. Int Orthop, Apr 2017, 41(4), 797-804. (Clinical study)
- SCHAGEMANN, J., et al. Mid-term outcome of arthroscopic AMIC for the treatment of articular cartilage defects in the knee joint is equivalent to mini-open procedures. Arch Orthop Trauma Surg, Jan 22 2018. (Clinical study)
- STEINWACHS, M.R.,et al. Systematic Review and Meta-Analysis of the Clinical Evidence on the Use of Autologous Matrix-Induced Chondrogenesis in the Knee. Cartilage. 2019:1947603519870846. https://www.ncbi.nlm.nih.gov/ pubmed/31508990 (Review of clinical studies)
- FOSSUM, V., et al. Collagen-Covered Autologous Chondrocyte Implantation Versus Autologous Matrix-Induced Chondrogenesis: A Randomized Trial Comparing 2 Methods for Repair of Cartilage Defects of the Knee. Orthopaedic Journal of Sports Medicine. 019;7(9):2325967119868212. doi.org/10.1177/2325967119868212 (Clinical study)