Geistlich Bio-Gide®
User Benefits
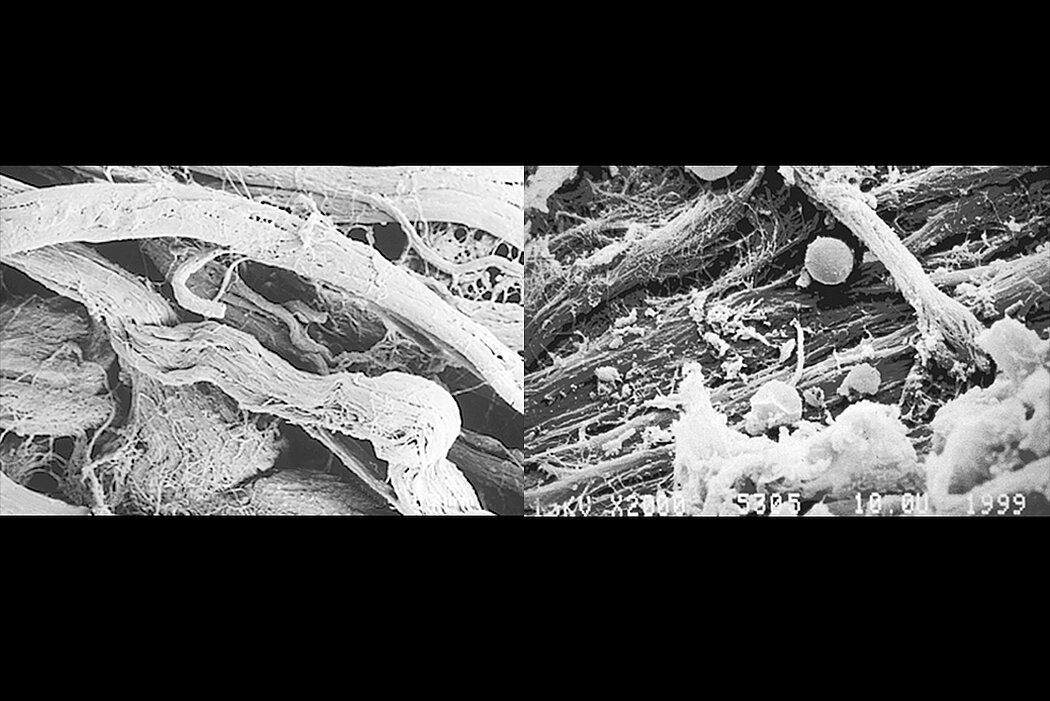
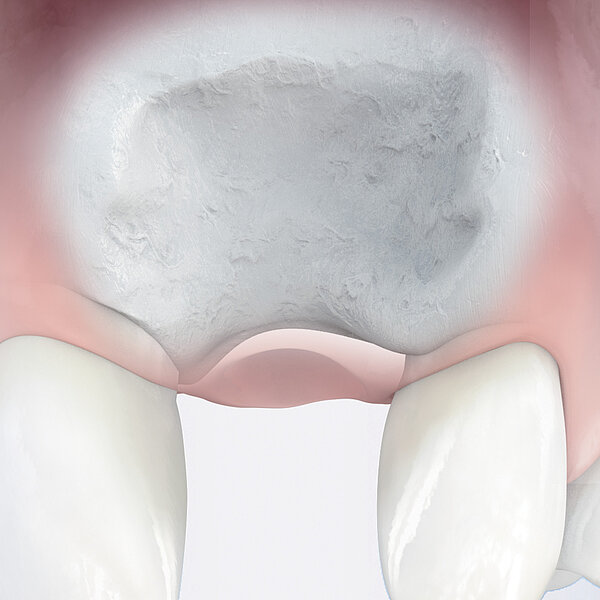
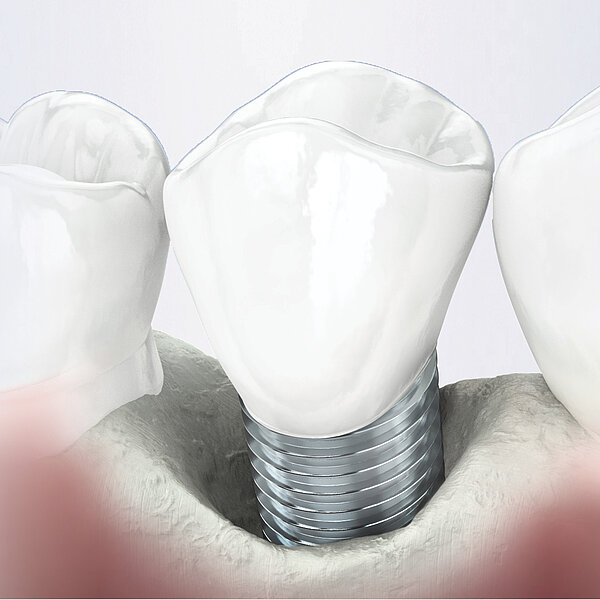
Geistlich Bio-Gide® is a collagen membrane for reliable bone regeneration and optimal tissue integration.1-3. The natural collagen structure of Geistlich Bio-Gide® permits prompt and homogeneous vascularization and so brings about optimal tissue integration and wound stabilization.5 The smooth side of Geistlich Bio-Gide® prevents soft tissue from growing into the defect and serves as a scaffold for the attachment of fibroblasts. 1,4-7 The rough, open-pored layer side of Geistlich Bio-Gide® serves as a framework for bone cells and blood vessels. 1,9. Geistlich Bio-Gide® is used in combination with a bone substitute such as Geistlich Bio-Oss® or Geistlich Bio-Oss® Collagen.
References:
- Schwarz F et al. Clin. Oral Implants Res. 2006;17(4):403-409. (Pre-clinical study)
- Rothamel D et al. Clin. Oral Implants Res. 2005; 16(3): 369-378. (Pre-clinical study)
- Schwarz F et al. Clin Oral Implants Res. 2014 Sept;25(9):1010-5. (Clinical study)
- Tal H et al. Clin Oral Implants Res. 2008; 19(3) : 295-302. (Clinical study)
- Rothamel D et al. Clin. Oral Implants Res. 2005; 16(3): 369-378. (Pre-clinical study
- Zitzmann NU et al. Int J Oral Maxillofac Implants.12, 1997;844-852. (Clinical study)
- Rothamel D et al. Clin. Oral Implants Res. 2004;15:443-449. (Pre-clinical study)
- Becker J et al. Clin Oral Implants Res. 2009; 20(7):742-749. (Clinical study)
- Schwarz F et al. Clin. Oral Implants Res. 2008;19(4): 402-415. (Pre-clinical study
- Perelman-Karmon M et al. Int J Periodontics Restorative Dent. 2012 Aug;32(4):459-65. (Clinical study)
- Buser D et al. J Periodontol 2013; 84(11): 1517-27. (Clinical study).
- Jung RE et al. Clin. Oral Implants Res. 2013 Oct;24(10):1065-73. (Clinical study)
- Pubmed September 2016. Search Term: Bio-Gide.
- iData Research Inc., US Dental Bone Graft Substitutes and other Biomaterials Market, 2015.
- iData Inc., European Dental Bone Graft Substitutes and other Biomaterials Market, 2015
Leading clinicians rely on Geistlich Bio-Gide® for:
- Optimal tissue integration and wound stabilization1,5,9
- High therapy safety even in case of dehiscence 4,8
- 11% more new bone formation with Geistlich Bio-Oss® and Geistlich Bio-Gide® vs Geistlich Bio-Oss® alone10
- Simplified surgery without removal of the membrane
- Aesthetically stable results after 5-9 years11
- Long-term data over 12 to 14 years show predictable results of bone augmentations with Geistlich Bio-Oss® and Geistlich Bio-Gide®12
In the 1990s, Dr. Peter Geistlich was the first to develop a natural collagen membrane that optimally meets the requirements for regenerative dentistry. Meanwhile Geistlich Bio-Gide® is a collagen membrane1,2, validated by over 20 years of successful use in more than two million patients and documented in more than 250 scientific publications13-15.
Application

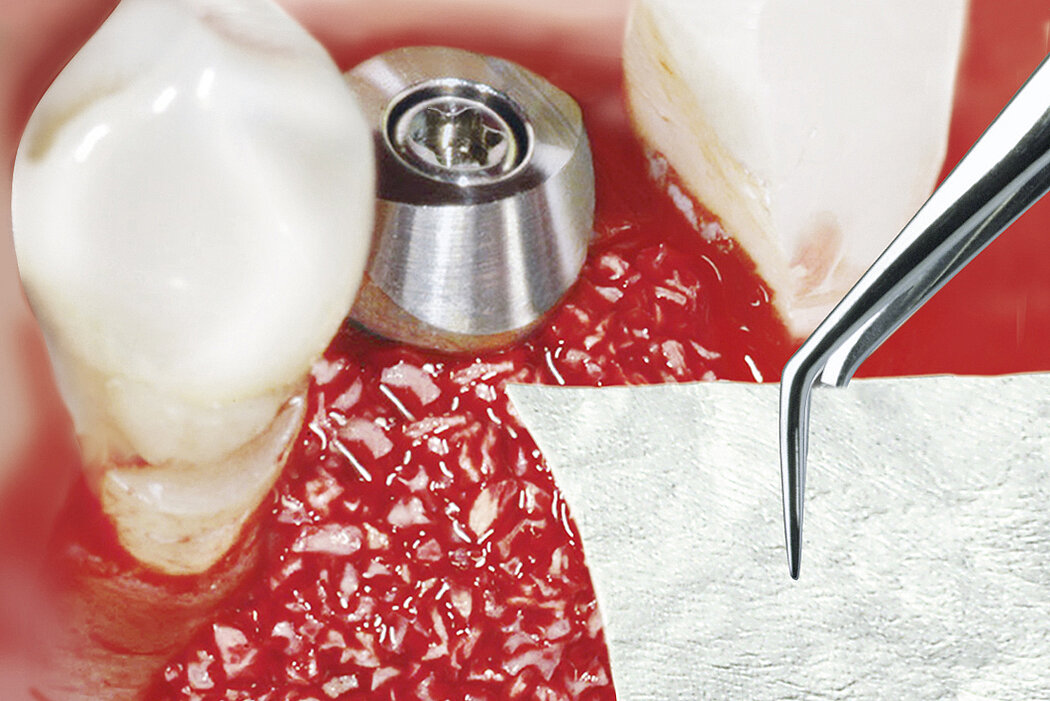
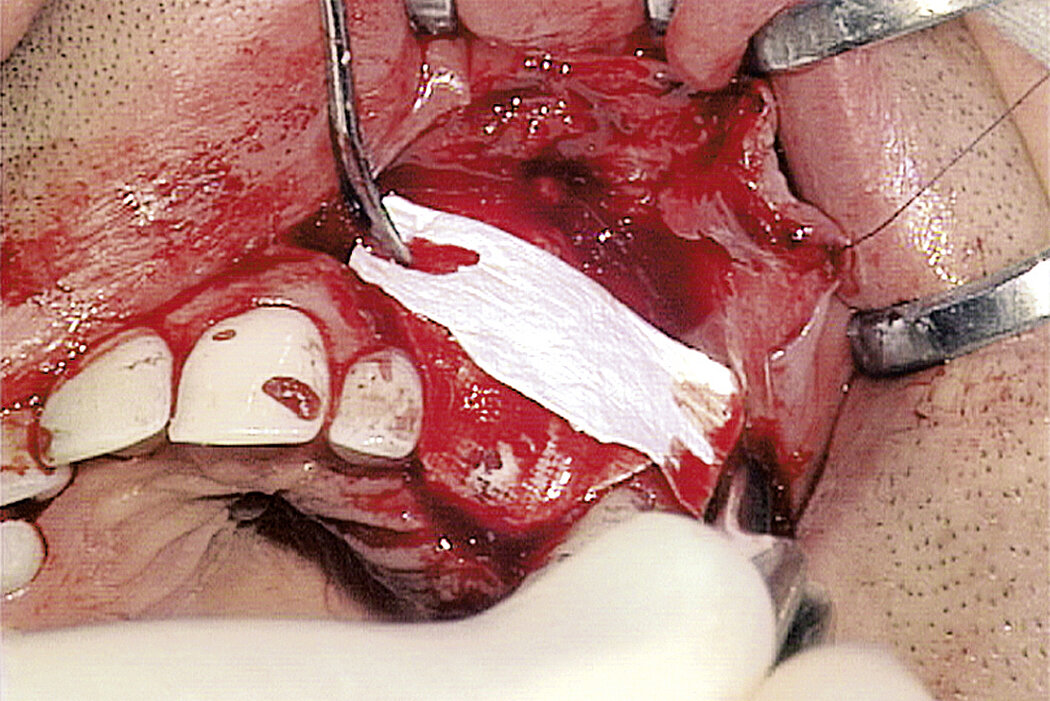
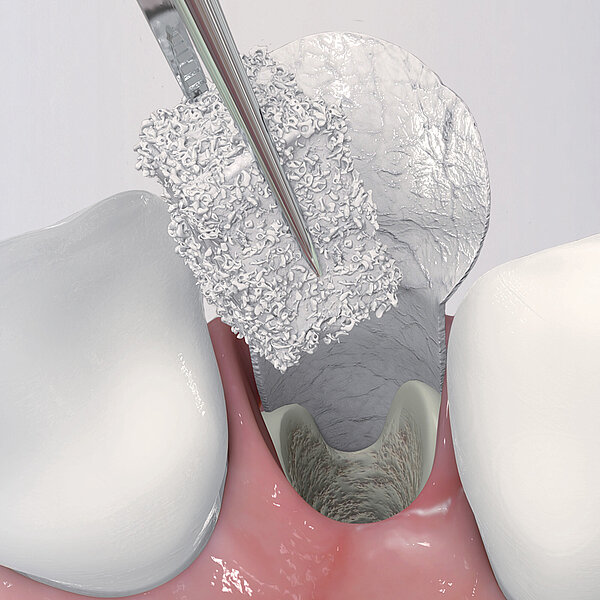
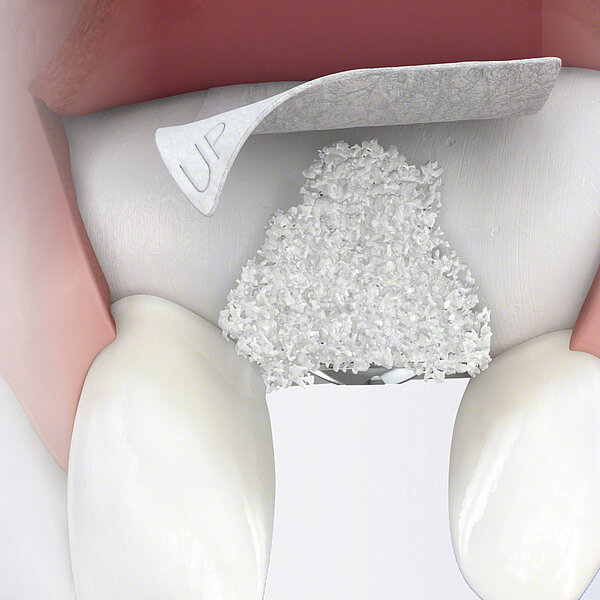
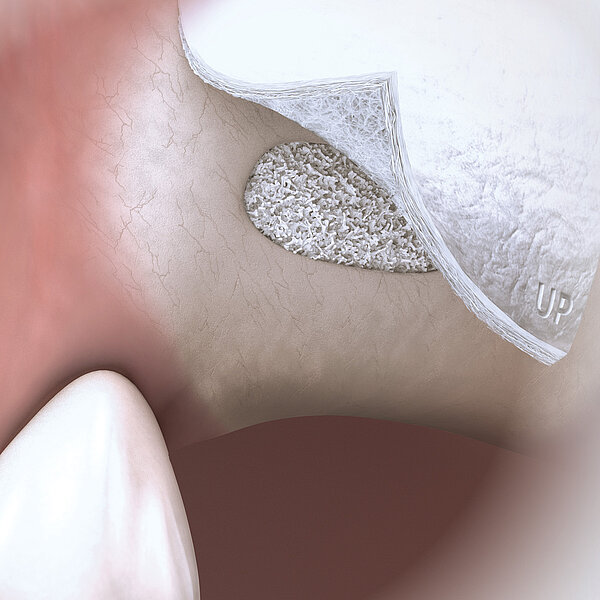
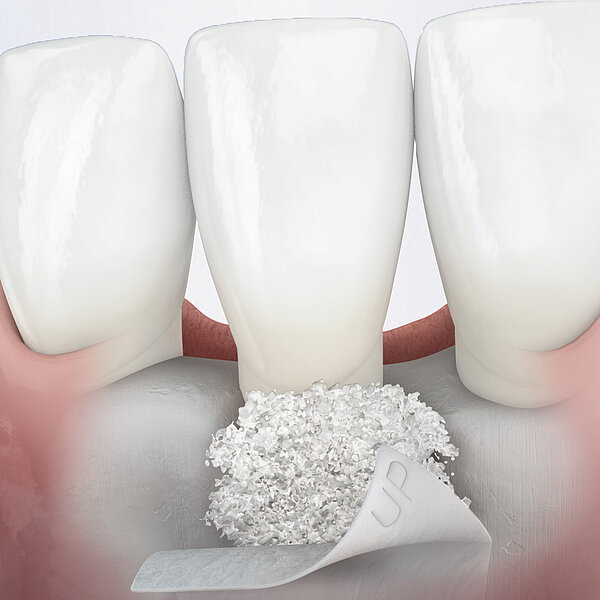
Geistlich Bio-Gide® should always be used with a bone substitute, such as Geistlich Bio-Oss® or Geistlich Bio-Oss® Collagen. Application of Geistlich Bio-Gide® membrane is easy:
- Measure the defect with a periodontal probe.
- Cut the dry collagen membrane to the desired shape and size.
- Apply dry with the rough side facing the defect.
- Moisten the membrane in situ with blood from the defect.
The membrane may be sutured or fixed with pins, but this is unnecessary in most cases due to its marked hydrophilic and adhesive properties.
References:
-
Data on File. Geistlich Pharma AG, Wolhusen, Switzerland. (Non-clinical)

Product Range
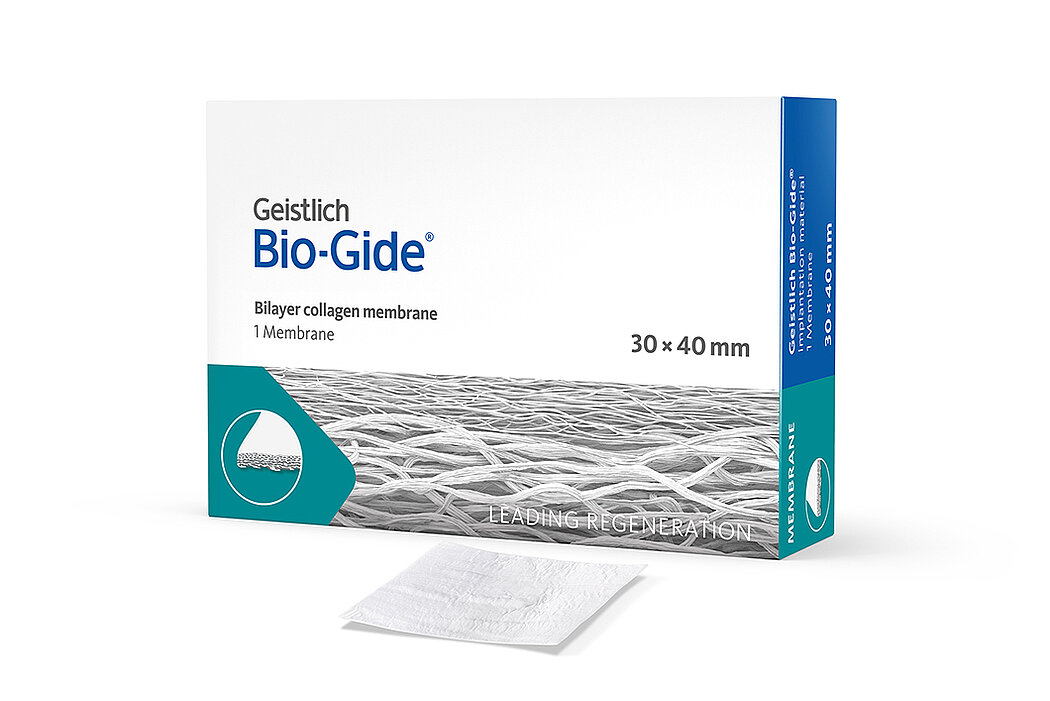
Geistlich Bio-Gide® is a unique bilayer collagen membrane made of porcine collagen.
Geistlich Bio-Gide® is available in three convenient sizes:
- 13 x 25 mm
- 25 x 25 mm
- 30 x 40 mm
(* Product availability may vary from country to country)
and a combination package:
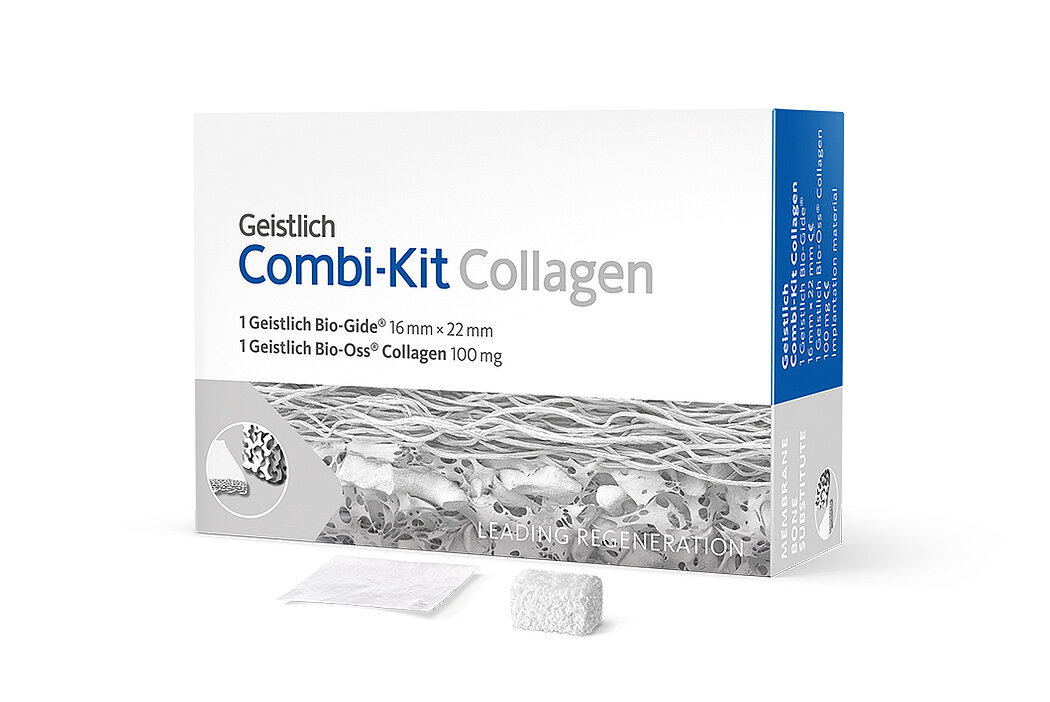
- Geistlich Combi-Kit Collagen
(Geistlich Bio-Gide® 16 x 22 mm with Geistlich Bio-Oss® 100 mg)
Not all products presented on this website are registered and approved for sale and usage in all countries or regions by the relevant authorities. Indications of use may also vary by country and region. Please contact your country representative of Geistlich Pharma AG for product availability and information.