Geistlich Bio-Oss® Collagen
User Benefits
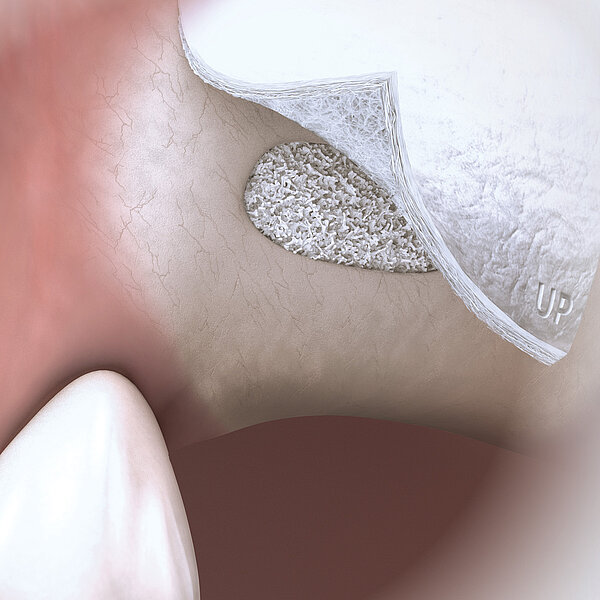
Geistlich Bio-Oss® Collagen consists of 90% Geistlich Bio-Oss® granules and 10% porcine collagen. The 90% of Geistlich Bio-Oss® particles provide Geistlich Bio-Oss® Collagen with all the advantages of the scientifically proven No.1 biomaterial in regenerative dentistry1,2.
The additional 10% of porcine collagen make it formable and easy to handle. Its regenerative potential clearly distinguishes Geistlich Bio-Oss® Collagen from mere collagen plugs.
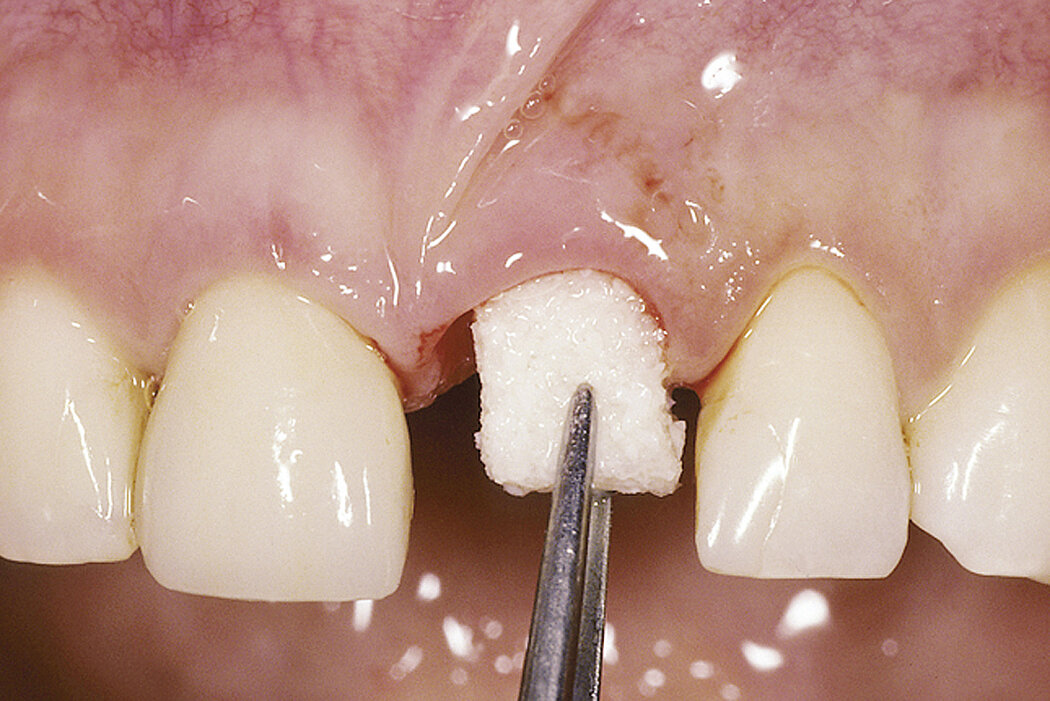
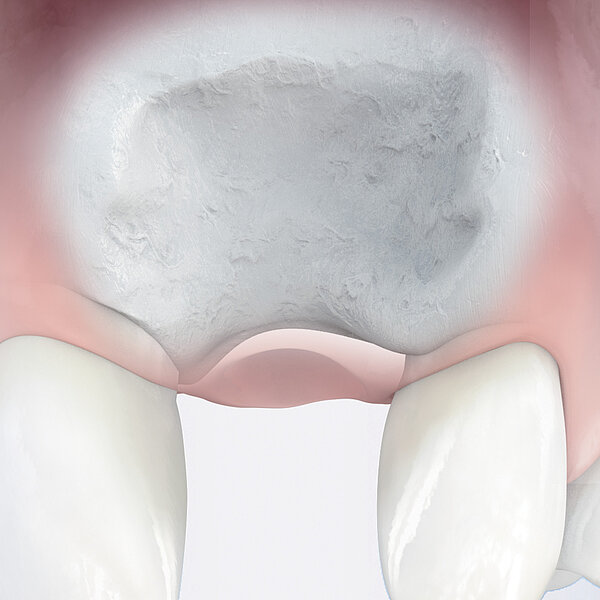
Geistlich Bio-Oss® Collagen is used in the most varied indications, including ridge preservation, minor bone augmentation and periodontal regeneration. The collagen is absorbed after a few weeks and does not replace the barrier function of a membrane.
References:
- Millennium Research Group, Dental Biomaterials North America, 2018 (Market research).
- Millennium Research Group, Dental Biomaterials Europe, 2018 (Market research).
- Trevisiol L et al., J Craniofac Surg. 2012 Sep;23(5):1343–8 (Clinical study).
- Rohner D et al., Int J Oral Maxillofac Surg. 2013 May;42(5):585-91 (Clinical study).
- Cardaropoli D et al., Int J Periodontics Restorative Dent. 2012 Aug;32(4):421–30 (Clinical study).
- Jung RE et al., J Clin Periodontol. 2013 Jan;40(1):90–8 (Clinical study).
- Araújo MG et al., Clin Oral Implants Res. 2010 Jan;21(1):55-64 (Preclinical study).
- Mordenfeld A et al., Clin. Oral Implant Res. 2010, Sep;21(9):961–70 (Clinical study). 13
- Sculean A et al., J Clin Periodontol. 2005 Jul;32(7):720-4 (Clinical study).
- Nevins ML et al., Int J Periodontics Restorative Dent. 2003 Feb;23(1):9-17 (Clinical study).
Geistlich Bio-Oss® Collagen

Geistlich Bio-Oss® Collagen is “the Master’s Choice” because:
- Added collagen in Geistlich Bio-Oss® Collagen improves handling and tailoring to the morphology of the defect3,4
- Geistlich Bio-Oss® particles serve as a scaffold for new bone and ensure predictable bone regeneration5,6
- Geistlich Bio-Oss® Collagen augmented tissue remains volume-stable in the long term due to the low resorption rate of the material7,8
- Geistlich Bio-Oss® Collagen significantly improves clinical attachment and pocket depth in periodontal surgery9
- Geistlich Bio-Oss® Collagen has the capacity to enable regeneration of the periodontal attachment apparatus in intrabony defects10
Handling characteristics are enhanced through the addition of 10% collagen. Geistlich Bio-Oss® Collagen block is easily modelled and adheres well to the defect site.
References:
- Millennium Research Group, Dental Biomaterials North America, 2018 (Market research).
- Millennium Research Group, Dental Biomaterials Europe, 2018 (Market research).
- Trevisiol L et al., J Craniofac Surg. 2012 Sep;23(5):1343–8 (Clinical study).
- Rohner D et al., Int J Oral Maxillofac Surg. 2013 May;42(5):585-91 (Clinical study).
- Cardaropoli D et al., Int J Periodontics Restorative Dent. 2012 Aug;32(4):421–30 (Clinical study).
- Jung RE et al., J Clin Periodontol. 2013 Jan;40(1):90–8 (Clinical study).
- Araújo MG et al., Clin Oral Implants Res. 2010 Jan;21(1):55-64 (Preclinical study).
- Mordenfeld A et al., Clin. Oral Implant Res. 2010, Sep;21(9):961–70 (Clinical study). 13
- Sculean A et al., J Clin Periodontol. 2005 Jul;32(7):720-4 (Clinical study).
- Nevins ML et al., Int J Periodontics Restorative Dent. 2003 Feb;23(1):9-17 (Clinical study).
Application
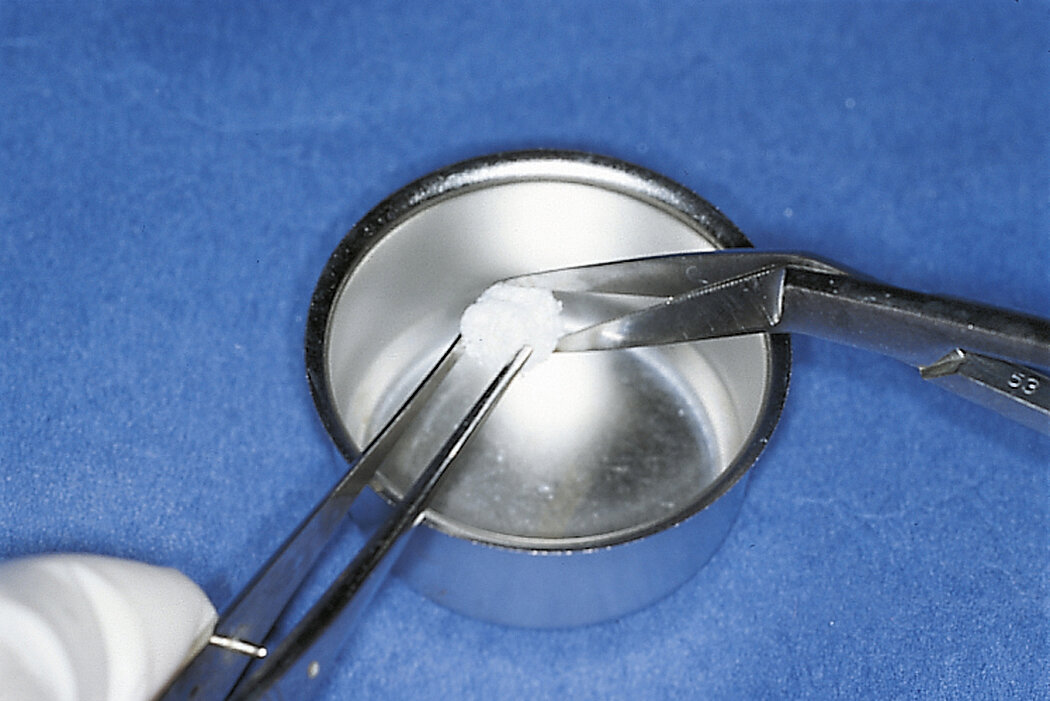
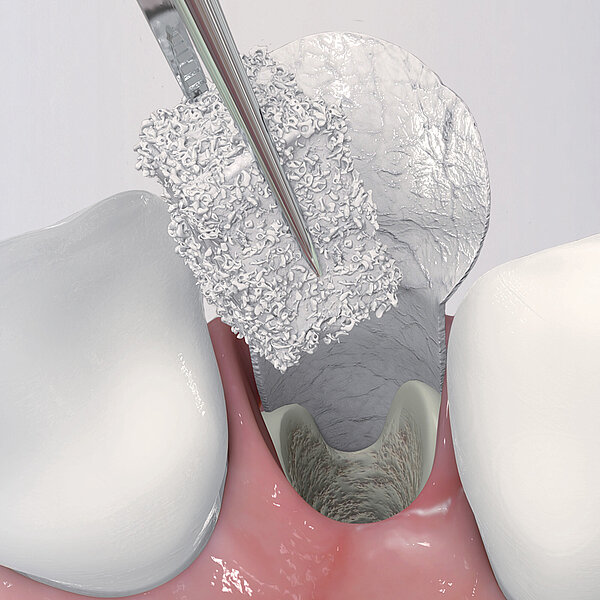
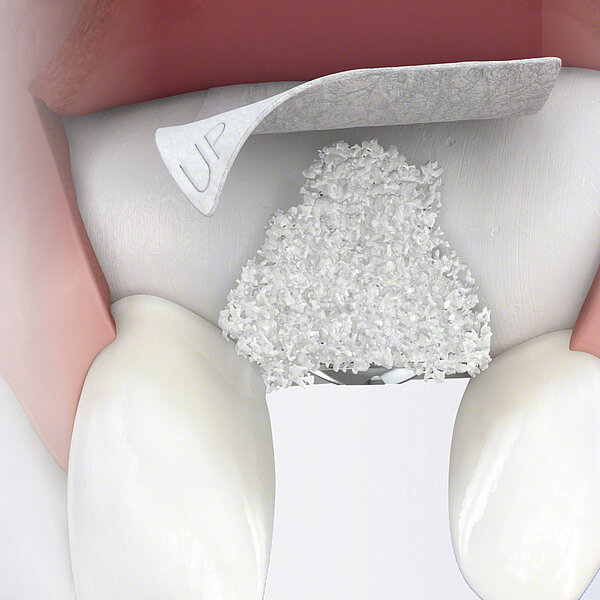
Preparation of Geistlich Bio-Oss® Collagen is easy:
- Cut the Geistlich Bio-Oss® Collagen block to the appropriate size
- Apply it dry, or moisten with saline solution
- When moistened, mould to the desired shape
- Place in defect site with forceps
- Avoid excessive compression
Product Range
Geistlich Bio-Oss® Collagen contains 90% small Geistlich Bio-Oss® granules and 10% porcine collagen. The small amount of collagen improves the handling capacities and the bone substitute material encourages effective and reliable bone formation.
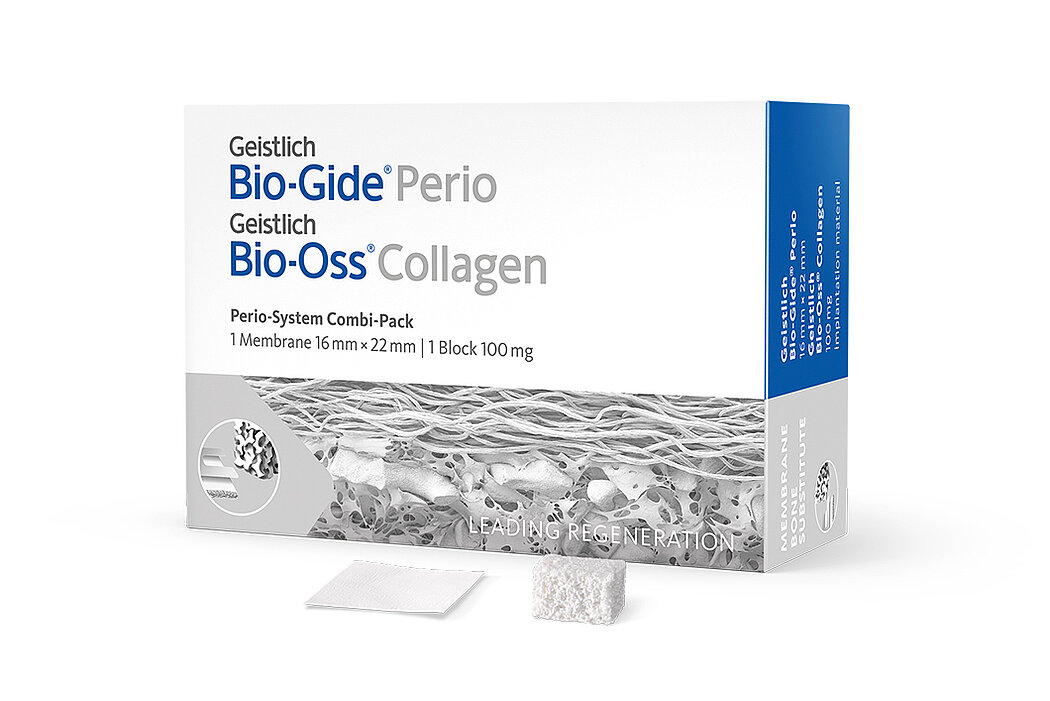
Geistlich Bio-Oss® Collagen is available in four block sizes:
- 50mg* (approx. 2.5mm x 5.0mm x 7.5mm)
- 100mg (approx. 5.0mm x 5.0mm x 7.0mm)
- 250mg (approx. 7.0mm x 7.0mm x 7.0mm)
- 500mg (approx. 10.0mm x 10.0mm x 7.0mm)
*Product availability may vary from country to country
and two combination packages:
- Geistlich Combi-Kit Collagen (with Geistlich Bio-Gide®)
- Perio-System Combi-Pack (with Geistlich Bio-Gide® Perio)
Not all products presented on this website are registered and approved for sale and usage in all countries or regions by the relevant authorities. Indications of use may also vary by country and region. Please contact your country representative of Geistlich Pharma AG for product availability and information.